Europe Anti Snoring Devices Market Size, Share, Trends & Growth Forecast By Device, Surgical Procedure and Country (UK, France, Spain, Germany, Italy, Russia, Sweden, Denmark, Switzerland, Netherlands, Turkey, Czech Republic and Rest of Europe) - Industry Analysis, From (2025 to 2033)
Europe Anti Snoring Devices Market Size
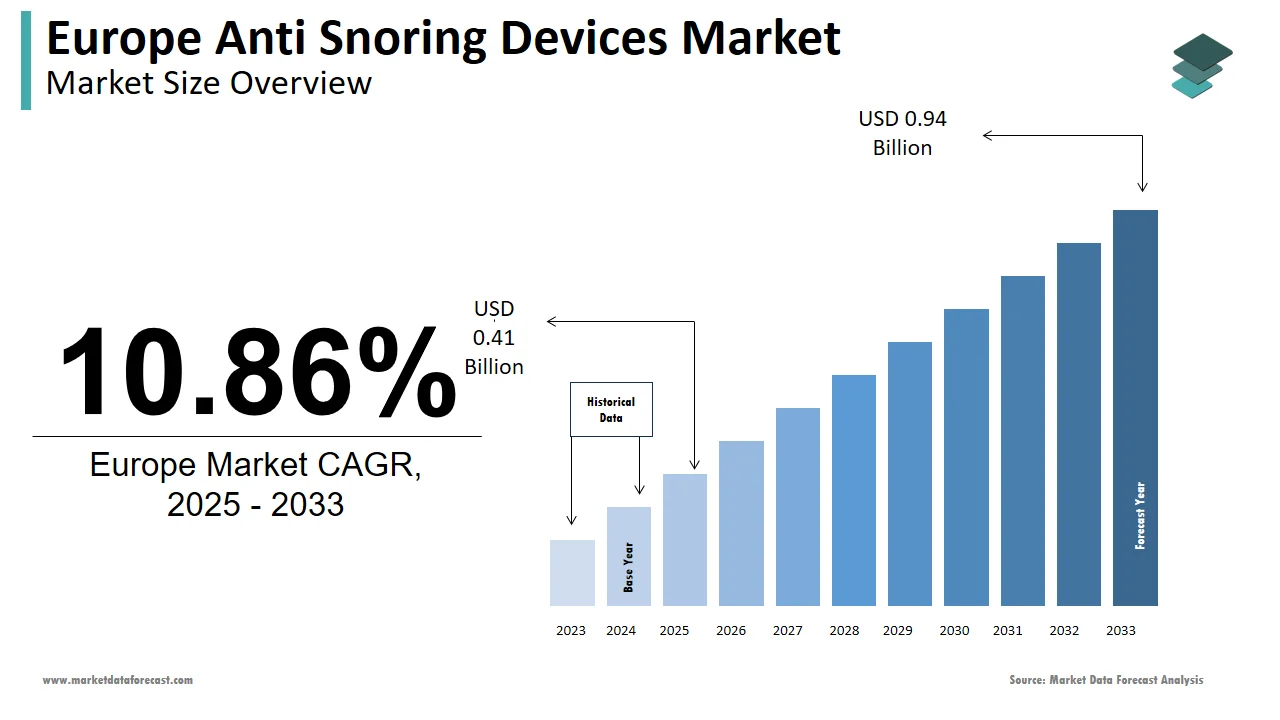
The anti-snoring devices market size in Europe was valued at USD 0.37 billion in 2024. The European market is estimated to be worth USD 0.94 billion by 2033 from USD 0.41 billion in 2025, growing at a CAGR of 10.86% from 2025 to 2033.
MARKET DRIVERS
Health Risks and Surgical Treatments Driving Demand for Anti-Snoring Solutions
Snoring is a common sleep disorder resulting from the repetitive narrowing and collapsing of the upper airway. Untreated snoring is often associated with adverse health outcomes, including systemic hypertension, coronary artery disease, stroke, and atrial fibrillation. In addition, snoring is also considered a significant social problem and leads to a more reduced quality of life for bed partners through disrupted sleep. Treatment for snoring involves either anti-snoring devices or doing an appropriate surgery. Conventional surgery includes uvulopalatopharyngoplasty surgery, tonsillectomy and adenoidectomy surgery, maxillo-mandibular and genioglossus advancement surgeries, radiofrequency ablation surgery.
A few of the key factors promoting the anti-snoring devices market in the European region are the increase in the awareness among individuals regarding the ill effects of snoring, soaring geriatric and obese population, the presence of an untapped population pool characterized by a snoring condition, and the increasing consumption of alcohol and cigarettes. The subsequent health risks associated with this condition are sleep apnea, interrupted breathing, and strain on the heart. These conditions have stimulated the population to opt for treatment solutions, including non-surgical appliances and surgical options, which has led to the overall industry's growth.
MARKET RESTRAINTS
High Costs, Limited Treatment Efficacy, and Reimbursement Issues
Be that as it may, such as the high cost of these surgeries, limited efficacy of the current treatments available for this condition, and unpropitious reimbursement issues are likely to deter the growth of anti-snoring devices and the snoring surgery market. Moreover, disadvantages to the use of the non-surgical equipment, such as dry mouth, jaw discomfort, and hypersalivation, serve as solid deterrents that discourage their use among the patients.
REGIONAL ANALYSIS
Geographically, the European anti-snoring devices market held the second-largest share in the global market during the forecast period. Factors such as growth in the awareness among individuals regarding the detrimental effects of this condition, the presence of many players in this sector, and an untapped population suffering from this condition are a few of the key factors boosting the market’s growth rate.
In 2024, the UK accounted for the majority market share in the European anti-snoring devices market. High intake of alcohol and cigarettes may reduce the blood flow to the ear, creating hearing impairments. The prevalence of the factor, as mentioned earlier, is high in Europe, which is predicted to drive market growth in the United Kingdom. In addition, technologically advanced new devices and therapeutics of the anti-snoring devices market are further accelerating the development of the UK anti-snoring devices.
The French anti-snoring devices market is predicted to showcase a healthy CAGR due to increased intake of alcohol and cigarettes, unfavorable reimbursement policies, and high costs associated with the surgeries are majorly boosting the market growth. However, the custom-made oral devices may restrain the anti-snoring devices market growth. In addition, the elder people population is more in France followed by the UK, and an overweight population responsible for the snoring diseases and increasing awareness related to illness of snoring and related advantages is driving the market of anti-snoring devices growth.
KEY MARKET PLAYERS
A few of the prominent companies leading the Europe anti-snoring devices market profiled in the report are Sleeping Well LLC., Apnea Sciences Corporation, Tomed Dr. Toussaint GMBH, AccuMED Corp, Fisher & Paykel Healthcare, Apnea Sciences Corporation, ImThera Medical Inc., ResMed Inc., SomnoMed, Sleep Well Enjoy Life, Ltd, MEDiTAS Ltd, and Nasal Devices.
MARKET SEGMENTATION
This Europe anti-snoring devices market research report is segmented and sub-segmented into the following categories.
By Product Type
- Mandibular Advancement Devices (MADs)
- Tongue Retaining Devices (TRD)
- Nasal Dilator
- Chin Strap
- Position Control
- Pillow
- Tongue Stabilizing Device (TSD)
- EPAP
By Surgical Procedures
- Uvulopalatopharyngoplasty (UP3)
- Laser-Assisted Uvula Palatoplasty (LAUP)
- Radiofrequency Ablation (RFA)
- Sclerotherapy
- Pillar
- Others
By Country
- UK
- France
- Spain
- Germany
- Italy
- Russia
- Sweden
- Denmark
- Switzerland
- Netherlands
- Turkey
- Czech Republic
- Rest of Europe
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2000
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com