Europe Home Insemination Products Market Size, Share, Trends & Growth Forecast Report By Procedure, Product Type, and By Country (UK, France, Spain, Germany, Italy, Russia, Sweden, Denmark, Switzerland, Netherlands, Turkey, Czech Republic & Rest of Europe) – Industry Analysis and Forecast, 2026 to 2034
Europe Home Insemination Products Market Size
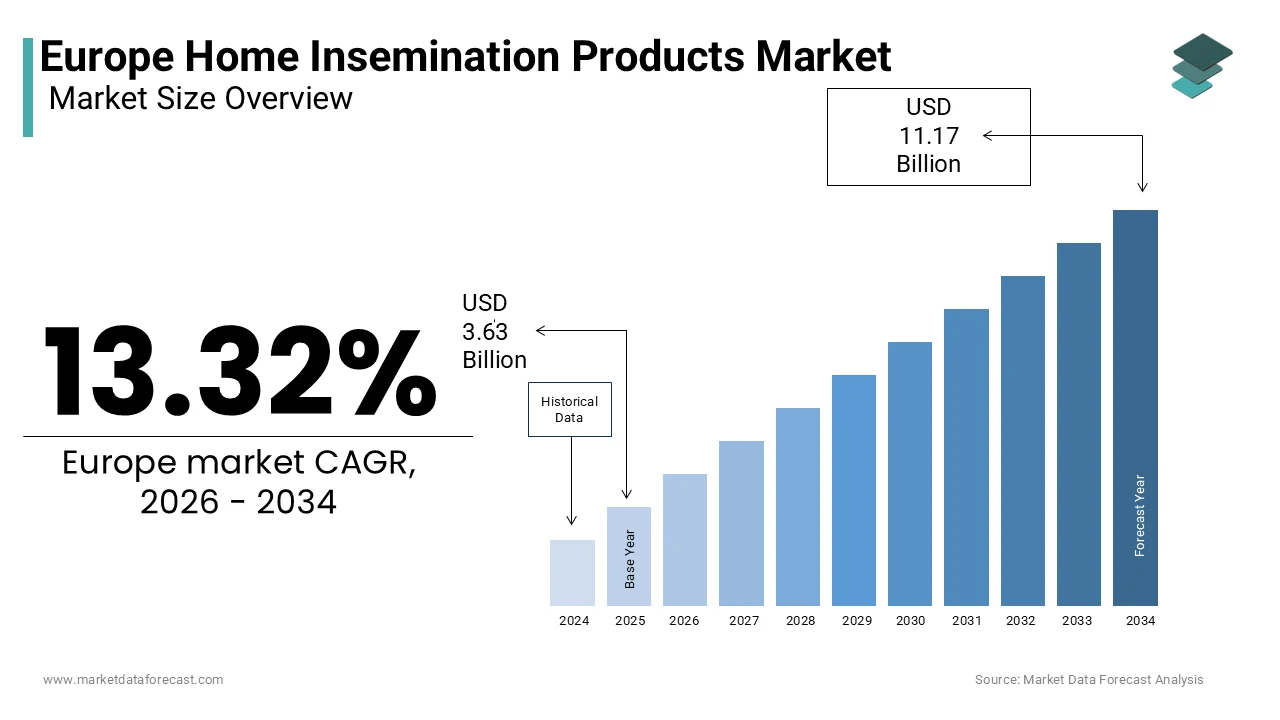
The Europe Home Insemination Products Market was valued at USD 3.63 billion in 2025, is estimated to reach USD 4.11 billion in 2026, and is projected to reach USD 11.17 billion by 2034, growing at a CAGR of 13.32% from 2026 to 2034.
MARKET OVERVIEW
Home insemination is a method of fertilization that contains sperm placed into the reproductive tract of the female with the help of a syringe. This method often suits single women, infertile couples, and lesbian couples. Some people are completely infertile, sub-fertile, and have unexplained infertility. Most couples experience difficulties in conceiving without undergoing fertility treatment. Such people would prefer home insemination before turning to the In-Vitro Fertilization (IVF) procedure. This procedure is done at home with the help of an insemination kit, which includes ovulation tests, semen containers, syringes, and other items to aid conception.
The European home insemination market is anticipated to grow during the forecast period due to driving factors like the growing rate of infertility in the area, changing lifestyles, and the growing preference of modern women for planned parenthood. Increasing support from the government through initiatives is ascribed to bolstering the demand of the local market. The factors affecting the home insemination market are mainly the strict regulations and firm laws in most of the Europe countries. Developing nations face the challenge of the absence of insurance coverage for fertility treatments, which further restricts the implementation of intrauterine insemination devices because of excessive out-of-pocket spending.
Impact of COVID-19 on European home insemination products market
The Coronavirus pandemic has influenced the IUI devices market moderately, with limitations on elective healthcare procedures. Companies are pushed to shut down or limit production processes for non-essential applications. Due to the health risk for pregnancies amid the lockdown restrictions and crisis, the demand for IUI devices has been further impacted severely. These aspects, along with a slump in consumer expenditure, have restricted investments and thus impacted the market size. However, the market is probably going to show recovery by 2021, with steady ease of lockdown. Following government measures (particularly stay-at-home orders and social distancing norms), doctors are postponing elective surgeries (unless critical) to prevent the spread of the virus to individuals with chronic or comorbidities conditions. Further, supply chain disruptions and movement restrictions have created a logistical nightmare for market players, resulting in severe product shortages in the European market.
This research report on the Europe home insemination products market has been segmented and sub-segmented based on the following categories.
Europe Home Insemination Products Market By Procedure
- Intracervical Insemination
- Intrauterine Insemination
- Intratubal Insemination
Based on the procedure, the intrauterine insemination segment is estimated to hold the highest share of the European market during the forecast period owing to the growing awareness of the different treatment procedures for fertility. Intrauterine insemination is a non-invasive procedure that is expected to increase the expansion of the regional home insemination market.
Europe Home Insemination Products Market By Product Type
- Home Conception Devices
- Insemination Kits
- Accessories
The insemination kits segment is estimated to be the fastest-growing segment in terms of value because of the growing geriatric population and is predicted to generate higher revenue during the forecast period.
Europe Home Insemination Products Market By End-user
- Fertility centers
- Hospitals
- Clinics
- Home care settings
- Others
The fertility centers segment is estimated to carry the largest share of the local market during the forecast period. Further, most pharmaceutical companies are working on developing new insemination kits that possess conventional benefits. This is expected to drive the expansion of the home insemination market.
Europe Home Insemination Products Market By Country
- UK
- France
- Spain
- Germany
- Italy
- Russia
- Sweden
- Denmark
- Switzerland
- Netherlands
- Turkey
- Czech Republic
- Rest of Europe
Europe's market is predicted to register a significant rate of growth during the forecast period.
This will be attributed to the unhealthy lifestyle and rising prevalence of infertility. Moreover, delays in pregnancy and changing lifestyles are the key factors driving the expansion of the artificial insemination market in Europe. Growing demand for Home Insemination by single mothers and lesbian couples, increasing prevalence of infertility among adults, and increasing key developments by market players have helped in the growth of the European home Insemination Market infrastructure.
KEY MARKET PLAYERS
Vitrolife, Pride Angel, Hi-Tech Solutions, Rocket Medical plc, Invo bioscience., Genea Limited and Rinovum Women's Health are some of the notable companies in the Europe home insemination products market.
RECENT HAPPENINGS IN THE MARKET
- In July 2019, FUJIFILM Irvine Scientific announced its new plans to construct a new cell culture media manufacturing site in Tilburg, Netherlands. This is to accelerate development in bioproduction and cell and gene therapy markets.
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2000
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com