North America Radiopharmaceuticals Market Research Report - Segmented By SPECT, PET, Application & Country (the United States, Canada, & Rest of North America) - Industry Analysis on Size, Share, Trends and Growth Forecast (2025 to 2033)
North America Radiopharmaceuticals Market Size
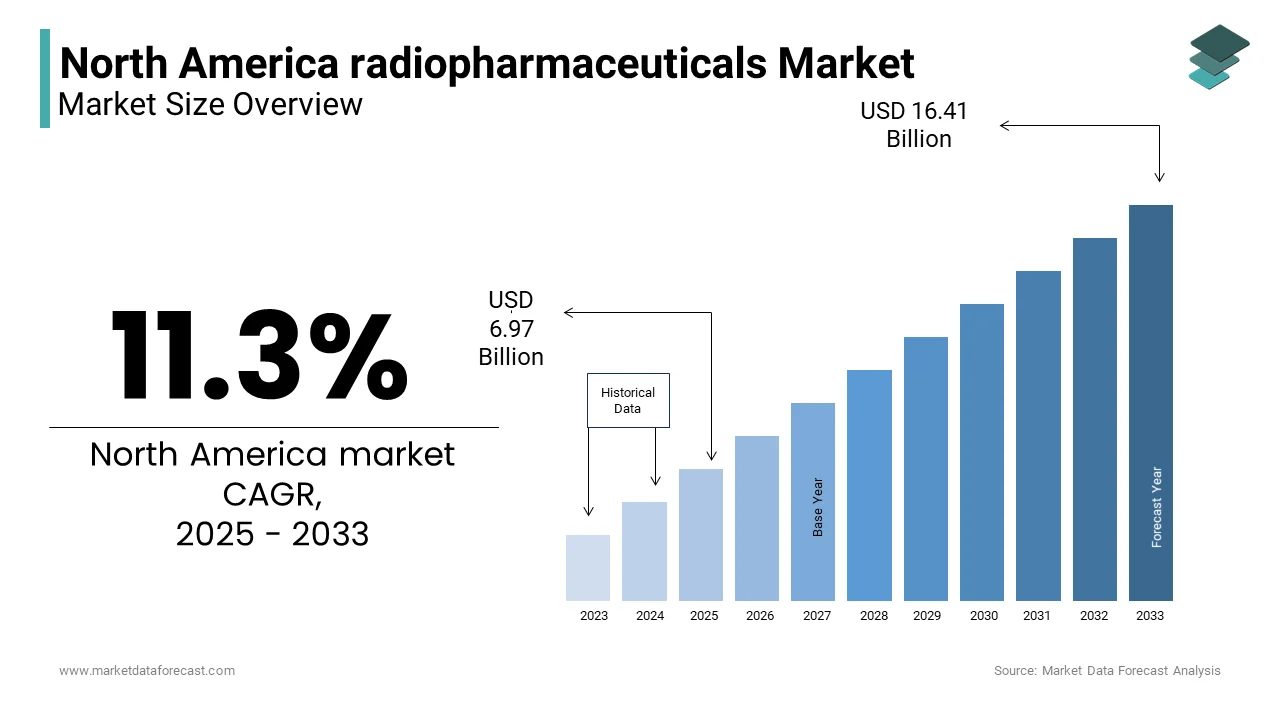
The North America radiopharmaceuticals Market was valued at USD 6.26 billion in 2024. The North America radiopharmaceuticals Market is expected to have 11.3% CAGR from 2025 to 2033 and be worth USD 16.41 billion by 2033 from USD 6.97 billion in 2025.
MARKET DRIVERS
The growing population diagnosed with cancer in the North American region is one of the major factors promoting the radiopharmaceuticals market in North America. There are 2 million new cancers reported in the United States. Cancer is a dangerous disease that leads to death in the short term. Therefore, analysis of which part of the body is affected by cancer cells is essential for the treatment procedure. For this purpose, radiopharmaceuticals has come into account. radiopharmaceuticals imaging for cancer is used for diagnosis, planning, and evaluating patient response to treatment. In this process, the researcher found several chemicals absorbed by specific organs and decided which organ in the body affects the cancer cells. Increasing awareness about this procedure among people suffering from different types of cancer and taking respective treatments for cancer drives the radiopharmaceuticals market forward.
In addition, factors such as technological advancements, the growing adoption of minimally invasive procedures, rising demand for medical imaging procedures by the increasing aging population across North America propel the regional market growth. The growing healthcare expenditure, increasing usage of radiopharmaceuticals in various applications such as diagnosis, staging, and treatment monitoring, an increasing number of initiatives such as Medicare and Medicaid to offer reimbursement for radiopharmaceuticals procedures by the North American governments and the availability of skilled professionals such as radiopharmaceuticals technologists and radiologists contributes to the growth of the North American radiopharmaceuticals market. Furthermore, the growing number of strategic partnerships and collaborations between healthcare organizations and radiopharmaceuticals companies and increasing awareness about the benefits of radiopharmaceuticals among people boost the growth rate of the regional market.
MARKET RESTRAINTS
However, one factor that restrains the growth of the radiopharmaceuticals market in this region is the lack of essential equipment for radiation protection and research facilities in some regions. Other factors, such as the high cost of the procedures, lack of specialized personnel, and lack of awareness about the procedure, hamper the market growth during the forecast period. In addition, radiation may occur health risks to some people who are sensitive to the radiation, and for some people, it can temporarily reduce bone marrow counts. Health risks for some people due to this radiopharmaceuticals, side effects after exposure to the radiation used for the procedure, and increasing alternative therapy hinder the market growth in this region.
REPORT COVERAGE
|
REPORT METRIC |
DETAILS |
|
Market Size Available |
2024 to 2033 |
|
Base Year |
2024 |
|
Forecast Period |
2025 to 2033 |
|
CAGR |
11.3 % |
|
Segments Covered |
By Type, Application and Country. |
|
Various Analyses Covered |
Global, Regional & Country Level Analysis, Segment-Level Analysis; DROC, PESTLE Analysis, Porter's Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
|
Country Covered |
The U.S., Canada and Rest of North America |
|
Market Leader Profiled |
Cardinal Health, Inc., Bracco Imaging S.p.A, G.E. Healthcare, Eczacibasi-Monrol Nuclear Products, Nordion, Inc. |
SEGMENTAL ANALYSIS
COUNTRY LEVEL ANALYSIS
North America had the highest share of the global market in 2023 and the domination of the North American market throughout the forecast period. The U.S. accounted for the largest share of the North American market in 2023 and is expected to register a promising CAGR during the forecast period owing to the growing adoption of advanced technologies and the introduction of new radiopharmaceuticals for diagnosis and therapy in the United States. The U.S. government is taking initiatives for cost-effective radiopharmaceuticals procedures and providing various training programs for developing radiopharmaceuticals for different diseases. As a result, the number of radiopharmaceuticals procedures in the United States has recently increased. The cardiology sector has the most growth due to the radiopharmaceuticals used in this region. The oncology sector has maximum growth due to this medicine usage. The number of positron emission tomography procedures is increasing in the United States is nearly 1.5 million per year.
Canada is another noteworthy regional market for radiopharmaceuticals in the North American market and is expected to register a healthy CAGR during the forecast period. Factors such as the growing prevalence of chronic diseases, such as cancer and cardiovascular diseases, the aging population, technological advancements in radiopharmaceuticals imaging, and government initiatives to promote healthcare access and reimbursement for radiopharmaceuticals procedures propel the Canadian market growth.
KEY MARKET PLAYERS
Companies such as Cardinal Health, Inc., Bracco Imaging S.p.A, G.E. Healthcare, Eczacibasi-Monrol Nuclear Products, Nordion, Inc., Bayer Healthcare, Lantheus Medical Imaging, Inc., Advanced Accelerator Applications S.A., IBA Molecular Imaging and Mallinckrodt PLC. are some of the major companies in the North America radiopharmaceuticals market.
MARKET SEGMENTATION
This research report on the North American radiopharmaceuticals market has been segmented and sub-segmented into the following categories
By Type
- SPECT
- PET
By Application
- Cardiology
- Lymphoma
- Thyroid
- Neurology
- Oncology
- others
By Country
- United States
- Canada
- Rest of North America
Frequently Asked Questions
What was the size of the North American nuclear medicine market in 2024?
The North American nuclear medicine market was valued at USD 6.26 bn in 2024.
What is the growth rate of the North American nuclear medicine market?
The North American nuclear medicine market is expected to grow at a CAGR of 11.3% from 2025 to 2033.
Who are the major players in the North American nuclear medicine market?
Cardinal Health, Inc., GE Healthcare, Siemens Healthineers, and Curium Pharmaceuticals are some of the major players in the North American nuclear medicine market.
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from $ 2000
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: [email protected]
