Asia Pacific Endoscope Reprocessing Market Size, Share, Trends & Growth Forecast Report By Product, End User and Country (India, China, Japan, South Korea, Australia, New Zealand, Thailand, Malaysia, Vietnam, Philippines, Indonesia, Singapore & Rest of APAC) - Industry Analysis From (2025 to 2033)
Asia Pacific Endoscope Reprocessing Market Size
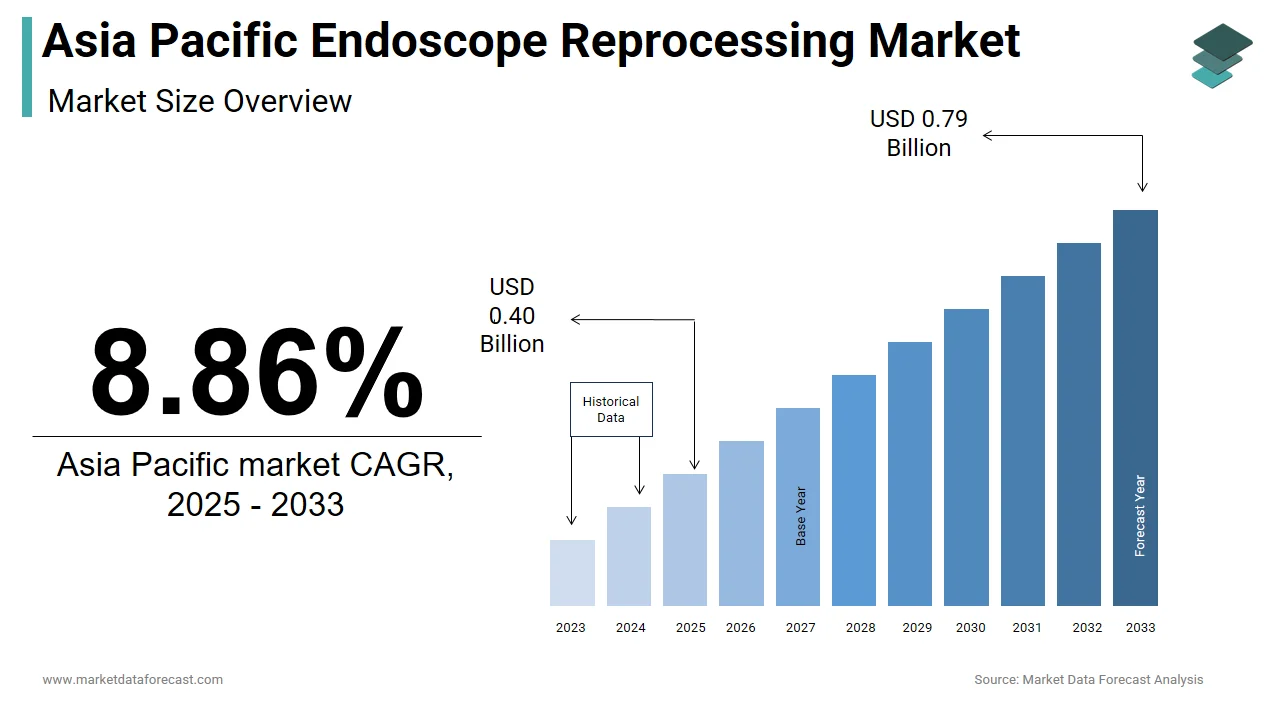
The size of the Asia Pacific endoscope reprocessing market was worth USD 0.37 billion in 2024. The Asia Pacific market is anticipated to grow at a CAGR of 8.86% from 2025 to 2033 and be worth USD 0.79 billion by 2033 from USD 0.40 billion in 2025.
MARKET SCENARIO
The Asia Pacific market is developing due to the rising popularity of endoscopic procedures, the high risk of endoscope-associated infections, the growing senior population, and the growing importance of understanding reprocessing procedures by healthcare facilities.
In addition, rising demand for surgical treatments that result in faster recovery, fewer post-surgery infections, less scarring, minor discomfort, better bleeding control, and higher precision is predicted to drive up demand for various endoscopic equipment. Furthermore, this country considers heavily investing in research and development, and the manufacturing of new drugs and medical devices creates market opportunities.
The market for endoscope reprocessors is likely to rise as technicians and physicians become more concerned about patient safety. Endoscope reprocessors are expected to drive demand for endoscope reprocessor systems due to their usage to prevent external and endogenous infections associated with flexible endoscopy.
Because of their composite nature and various uses in medical procedures, the risk of infection transmission associated with endoscopes is typically significant. As a result, health agencies have implemented rigid endoscope reprocessing regulations to improve security and reduce the spread of infections. Constant heating and cooling during disinfection procedures causes wear and tear on the insertion and light guiding tubes, reducing the equipment's durability.
Increased costs of endoscopy procedures and reprocessing equipment and limited reimbursements in this region, and growing concerns raised safety of reprocessed instruments are among the significant factors that will obstruct market growth and further challenge Asia pacific endoscope reprocessing market growth during the forecast period.
GEOGRAPHICAL ANALYSIS

Geographically, the Asia Pacific Endoscope Reprocessing Market is estimated to grow faster, resulting in a significant patient population in markets. In addition, public pressure to improve hospital care quality, the rising expense of HAIs in healthcare systems, the advent of multi-drug-resistant bacteria, and government initiatives contribute to the market's growth in these nations.
The India Endoscope Reprocessing Market is expected to develop due to rising endoscopy research support, increasing government investments, increasing the number of endoscopic surgeries and improving endoscopy infrastructure in India, and a growing number of hospitals in Singapore and South Korea. Endoscope reprocessing using single-shot disinfectants, rather than multi-use or reusable endoscope disinfectants, allows for effective and efficient cleaning of contaminated endoscopes.
The China Endoscope Reprocessing Market is predicted to reach a decent CAGR during the forecast period. As a result, no risk of cross-contamination occurs. As a result, an increasing number of endoscope reprocessor manufacturers are concentrating on producing single shot endoscope reprocessor models to provide high-quality services to various healthcare facilities throughout the regions. In addition, emerging economies in the Asia-Pacific area offer lucrative investment prospects for international infection control companies looking to relocate their operations there.
In September 2018, Ecolab inaugurated its Greater China Healthcare Innovation and Customer Experience Center in Shanghai. In October 2017, the company moved its headquarters to Singapore, expanding its operations across the APAC region. Expansions in emerging markets are encouraging endoscope reprocessing product use in the medical device manufacturing business in these nations, offering opportunities for product makers and service providers.
KEY MARKET PLAYERS
Notable companies operating in the APAC endoscope reprocessing market profiled in the report are Wassenburg Medical (The Netherlands), ARC Healthcare Solutions (Canada), and Metrex Research (US), Cantel Medical (US), Laboratories Anios (France), Olympus (Japan), Custom Ultrasonics (US), STERIS (US), Steelco (Italy), Getinge (Sweden), ENDO-TECHNIK (Germany), BES Decon (UK), Advanced Sterilization Products (ASP) (US).
MARKET SEGMENTATION
This Asia Pacific endoscope reprocessing market research report is segmented and sub-segmented into the following categories.
By Product
- High-Level Disinfectants & Test Strips
- Detergents & Wipes
- Automated Endoscope Reprocessors
- Single-Basin Automated Endoscope Reprocessors
- Double-Basin Automated Endoscope Reprocessors
- Endoscope Tracking Systems
- Advantages of Endoscope Tracking Systems
- Endoscope Drying, Storage, & Transport Systems
- Other Products
By End-User
- Ambulatory Surgery Centers
- Hospitals
- Other End Users
By Country
- India
- China
- Japan
- South Korea
- Australia
- New Zealand
- Thailand
- Malaysia
- Vietnam
- Philippines
- Indonesia
- Singapore
- Rest Of APAC
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2000
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com