Asia Pacific Erythropoietin Drugs Market Size, Share, Trends & Growth Forecast Report By Product, Application, Drug Class and Country (India, China, Japan, South Korea, Australia, New Zealand, Thailand, Malaysia, Vietnam, Philippines, Indonesia, Singapore & Rest of APAC) - Industry Analysis From (2025 to 2033)
Asia Pacific Erythropoietin Drugs Market Size
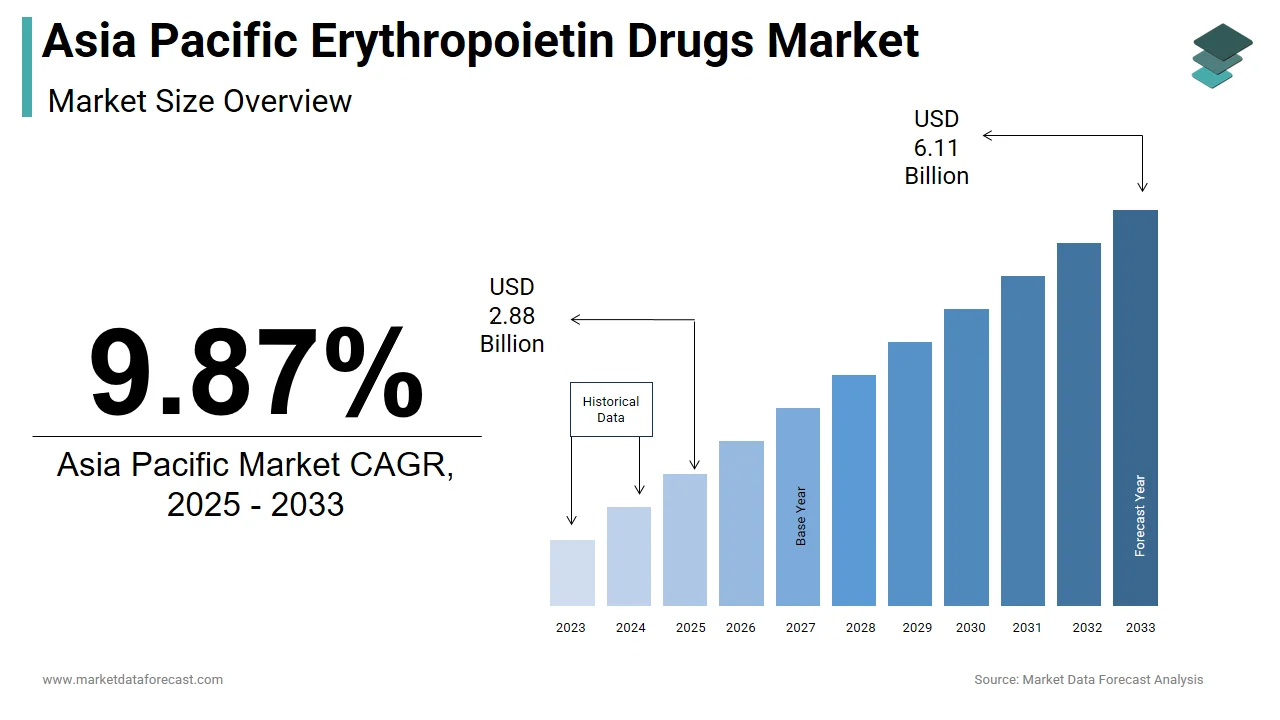
The size of the Asia Pacific erythropoietin drugs market was worth USD 2.62 billion in 2024. The Asia Pacific market is anticipated to grow at a CAGR of 9.87% from 2025 to 2033 and be worth USD 6.11 billion by 2033 from USD 2.88 billion in 2025.
MARKET DRIVERS
Rising Chronic Diseases and Demand for Biosimilars Fuel EPO Market Growth
The high prevalence of cancer, HIV and Chronic renal failure in developing APAC countries presents a significant opportunity for drug manufacturers to expand their presence in this market. The increased demand for low-cost therapies in developing regions in China, India, Taiwan and Pakistan etc. has prompted the development of biosimilars for EPO in these regions. These chronic diseases have the potential to cause anemia. To control the increasing cases of anemia, the demand for erythropoietin (EPO) drugs is expected to witness a surge in the forthcoming years.
The demand for erythropoietin (EPO) is expected to increase the R&D to give birth to newer medicines that have fewer side effects, in the coming years to combat the increasing incidence of anemia. The Erythropoietin (EPO) market is expected to grow during the forecast period owing to factors such as an increase in the number of patients with anemia, favorable reimbursements, increased commercialization of EPO biosimilars and an increased incidence of kidney diseases in APAC regions.
MARKET RESTRAINTS
Side Effects and Long Treatment Durations Hamper EPO Market Growth
Few factors restraining the market growth are side effects of EPO drugs, such as high blood pressure, iron deficiency, and influenza like illness, along with longer duration of EPO treatments mostly take 4-6 weeks are limiting market growth of erythropoietin-based drugs.
MARKET OPPORTUNITIES
Favorable Regulations and Marketing Trends Boost EPO Market Expansion
Growing marketing trends for erythropoietin biosimilars, readily available discounts, favorable government regulations, and increased awareness of the benefits of EPO therapies are all contributing to give new opportunities to the growth of the erythropoietin drug market.
MARKET CHALLENGES
Awareness about the availability of drugs in the market is challenging the market growth rate in APAC regions.
GEOGRAPHICAL ANALYSIS
Asia Pacific is a promising market for erythropoietin growth, and most erythropoietin companies are focused on the region. Due to the development of biosimilars by the major manufacturers in these regions, the China holds the largest share of the APAC EPO drugs market.
India and China are important markets for erythropoietin drugs industry. The Chinese market dominated the APAC EPO market by country in 2020, reaching a market value of $ 192 million. The Japanese market shows a CAGR of 16% at during (2025 - 2033). Additionally, the Indian market is expected to grow at a CAGR of 15% during (2025 - 2033). There is a great demand for low cost therapies in this region. As a result, Asia-Pacific has attracted many companies for the development of EPO drugs. They are focused on developing more drugs than anywhere else in the world.
Japan, on the other hand, will experience the fastest growing CAGR in the coming years. Another factor contributing to the growth of the market is the increase of government initiatives in this region to provide better healthcare services to the public. For example, the Japanese government has passed regulations requiring that biosimilars be reduced by 31% from their availability at subsidized prices to people of lower socioeconomic classes. In addition, India has the advantage of cheap labor and a strong pharmaceutical industry, these factors are expected to give good growth rate for the APAC regions.
KEY MARKET PLAYERS
The Top companies leading in the Asia Pacific erythropoietin drugs market profiled in the report are Johnson & Johnson, Celltrion, Inc, Teva Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd. , Dr. Reddy’s Laboratories Ltd.Amgen, Inc. , F. Hoffmann-La Roche Ltd. , LG Life Sciences Ltd. , Biocon Limited and Intas Pharmaceuticals Ltd.
MARKET SEGMENTATION
This Asia Pacific erythropoietin drugs market research report is segmented and sub-segmented into the following categories.
By Product
- Epoetin-alpha
- Epoetin-beta
- Darbepoetin-alfa
- Others
By Application
- Cancer
- Haematology
- Renal Diseases
- Neurology
By Drug Class
- Biologics
- Biosimila
By Country
- India
- China
- Japan
- South Korea
- Australia
- New Zealand
- Thailand
- Malaysia
- Vietnam
- Philippines
- Indonesia
- Singapore
- Rest Of APAC
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2000
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com