Global Autoimmune Monoclonal Antibodies Market Size, Share, Trends & Growth Analysis Report By Source, Application, End User and Region – Industry Forecast (2025 to 2033)
Global Autoimmune Monoclonal Antibodies Market Size
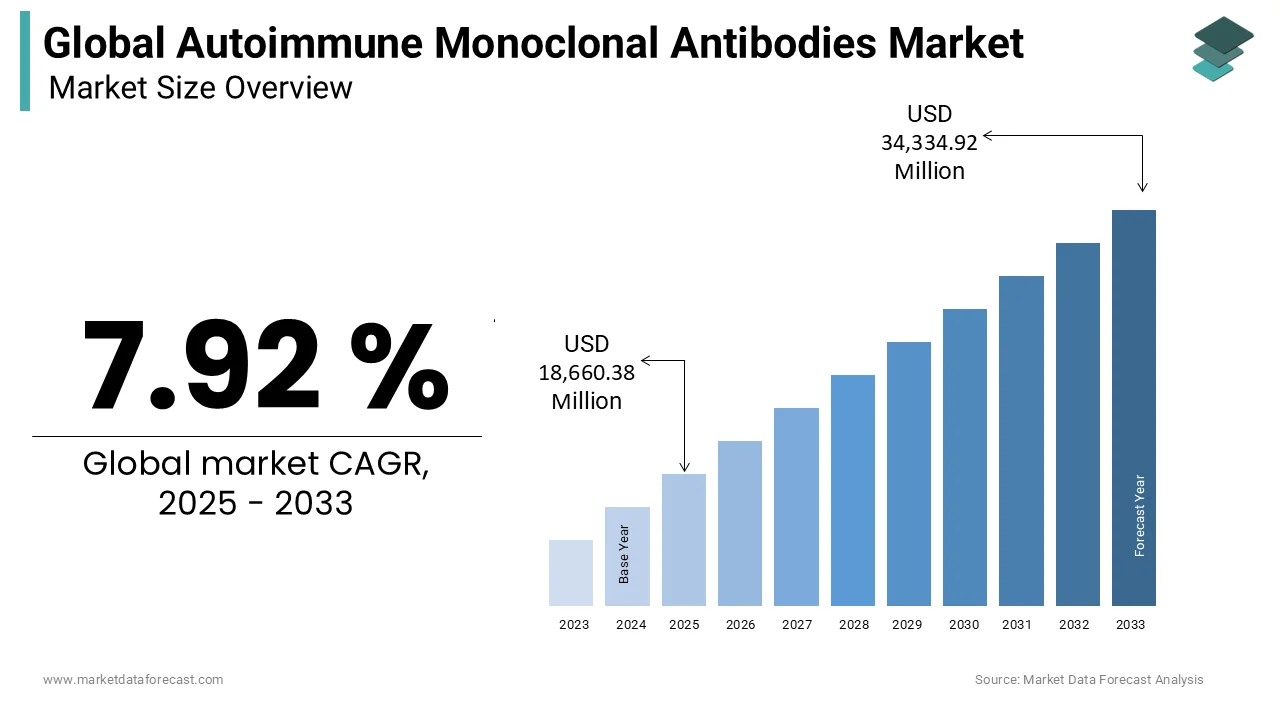
The global Autoimmune Monoclonal Antibodies Market was valued at USD 17,290.94 million in 2024, is estimated to reach USD 18,660.38 million in 2025, and is projected to reach USD 34,334.92 million by 2033, growing at a CAGR of 7.92% from 2025 to 2033.
MARKET DRIVERS
The growing number of projects and initiatives to spur the development of monoclonal antibodies, increase the number of patients with autoimmune diseases, and increase public & private organizations' involvement to support advanced therapeutics development driving the growth of the global autoimmune monoclonal antibodies market.
The rising prevalence of autoimmune disorders worldwide and developing markets drive this market's growth rate. The demand for autoimmune monoclonal antibodies is being propelled by increasing serious arthritis and multiple sclerosis cases. Along with this, the demand for autoimmune monoclonal antibodies has developed due to a rise in the family history of immune disease. The main factors are increased genomics research and development and the introduction of technologically innovative genetic platforms, such as next-generation sequencing. Because of their homogeneity, mAbs are perfect for laboratory procedures because they allow for similar antibodies. As a result, they can meet the growing demands of the healthcare industry.
Besides, the growing demand for antibody-based therapeutics, the rising adoption of advanced medical therapeutics, and the increasing number of lifestyle-related disorders further promote the growth of the global autoimmune monoclonal antibodies market. The growth of the global market is predicted to be driven by the increasing prevalence of cancer in the coming years. Increasing emphasis on genomics research and advancement and the advent of technologically innovative genetic platforms, such as next-generation sequencing, have all aided the industry. Furthermore, over the projected period, approval of mAbs for a range of indications is expected to raise utilization rates.
MARKET RESTRAINTS
The presence of stringent regulatory pathways, lack of advanced research facilities & underdeveloped healthcare infrastructure in certain developing regions, and limited accessibility of monoclonal antibody therapeutics due to high cost are restraining the autoimmune monoclonal antibodies market. One disadvantage of mAb-based medications is that they are highly selective and have small targets. This is because they only communicate with cells rather than penetrating them. This has hampered the global monoclonal antibody market to some extent. Furthermore, effective mAbs processing necessitates additional cell culture and purification procedures. Key players in the market are undertaking comprehensive studies to try to resolve these challenges.
REPORT COVERAGE
| REPORT METRIC | DETAILS |
| Market Size Available | 2024 to 2033 |
| Base Year | 2024 |
| Forecast Period | 2025 to 2033 |
| Segments Analysed | By Source, Application, End User, and Region |
| Various Analyses Covered | Global, Regional & Country Level Analysis, Segment-Level Analysis, DROC, PESTLE Analysis, Porter’s Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
| Regions Analysed | North America, Europe, Asia Pacific, Latin America, the Middle East, and Africa |
| Key market players
| GlaxoSmithKline plc (U.K.), AstraZeneca plc (U.K.), F. Hoffmann-La Roche Ltd |
SEGMENTAL ANALYSIS
By Source Insights
Wide usage of managing diseases such as cancer, inflammatory, and autoimmune diseases, humanized mAbs accounted for the highest revenue share. The use of advanced technologies, such as phage or yeast show and transgenic mice, for the generation of human antibodies is causing rapid growth in this industry. The first human antibody to be approved by the US was adalimumab.
By Application Insights
Due to rising rheumatoid arthritis incidences, accelerated uptake of biological therapies, and new product releases, the autoimmune disorder category is projected to develop.
By End-user Insights
In 2022, the hospital pharmacy segment is expected to be the top based on the end-user during the forecast period. Increased prevalence of chronic diseases fueling treatment rates, rising healthcare spending levels, and patient knowledge of treatment options are all projected to contribute to hospitals' dominance in the end-use segment over the forecast period.
REGIONAL ANALYSIS
Geographically, North America is expected to command the major market share of 45.0% of the global autoimmune monoclonal antibodies market in 2022 and is expected to grow at the highest CAGR of 8.47% during the forecast period. With recent technological advancements and several novel drug molecules to improve the care process, North America has the highest market share by percentage. Furthermore, increased federal support for cancer treatment and technological advancement is expected to accelerate therapeutic proteins' development. North America accounts for the largest market share by percentage due to the latest technology development and various innovative drug molecules to enhance the treatment procedure. Europe is considered the second-largest market for autoimmune monoclonal antibodies due to increasing government funding for autoimmune disease research and development.
Rising government support for autoimmune disease research and development has made Europe the second-largest demand for autoimmune monoclonal antibodies. Due to the disposable income, massive untapped capacity, increased research and development expenditure, and increasing patient awareness about the therapeutic use of mAbs, the Asia Pacific area is projected to expand significantly.
Asia-Pacific is predicted to account for the largest market share over the coming years for the autoimmune monoclonal antibodies market due to the risen pervasiveness of arthritis and related disorders and the number of generic drugs.
KEY MARKET PLAYERS
Some of the notable companies leading the global autoimmune monoclonal antibodies market profiled in this report are
- GlaxoSmithKline plc (U.K.)
- AstraZeneca plc (U.K.)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Bristol-Myers Squibb (U.S.)
- Johnson & Johnson (U.S.)
- Innovent Biologics, Inc. (China)
- Takeda Pharmaceutical Company Ltd. (Japan)
- Amgen Inc. (U.S.)
- Biogen Inc. (U.S.)
- UCB Company (Belgium).
RECENT HAPPENINGS IN THIS MARKET
- In October 2020, the US Food and Drug Administration (FDA) designated CARsgen's CT041 CLDN18.2 CAR-T cells as an orphan drug to combat gastric and gastroesophageal junction cancers.
- In October 2018, Amgen announced the introduction of Amgevita, a biosimilar to adalimumab, in Europe. Growing the number of treatment services open to the millions of people who suffer from chronic inflammatory diseases
- Imraldi, an adalimumab biosimilar, was launched by Biogen in October 2018.
- In October 2017 - Anti-PD-1 Monoclonal Antibody MGA012: Incyte and MacroGenics Announce a Global Collaboration and Licensing Agreement. Anti-PD-1 therapy is becoming a mainstay in cancer care across a wide range of tumor types, and we hope that adding MGA012 to our clinical pipeline would help us achieve our long-term immuno-oncology growth goals.
MARKET SEGMENTATION
This research report on the global autoimmune monoclonal antibodies market has been segmented and sub-segmented into the following categories.
By Source
-
Murine
- Chimeric
- Humanized
- Human
By Application
- Systemic Lupus Erythematosus
- Rheumatoid Arthritis
- Multiple Sclerosis
- Transplant Rejection/Graft Versus Host Disease
By End-user
- Hospitals/Clinics
- Research Institutes
- Diagnostic Laboratories
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- The Middle East and Africa
Frequently Asked Questions
What is the current size of the autoimmune monoclonal antibodies market?
The global autoimmune monoclonal antibodies market size is estimated to be USD 17290..94 Million in 2024.
What factors are driving the growth of the autoimmune monoclonal antibodies market?
The rising prevalence of autoimmune diseases, the growing demand for biologic therapies, and the development of new and innovative monoclonal antibody drugs majorly boost the growth rate of the autoimmune monoclonal antibodies market.
Who are the major players in the autoimmune monoclonal antibodies market?
AbbVie, Roche, Amgen, Johnson & Johnson, and Bristol Myers Squibb are some of the key players in the autoimmune monoclonal antibodies market.
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2500
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com