Europe Adhesion Barrier Market Size, Share, Trends & Growth Forecast Report By Product, Product Form, Surgical Application and Country (UK, France, Spain, Germany, Italy, Russia, Sweden, Denmark, Switzerland, Netherlands, Turkey, Czech Republic and Rest of Europe) - Industry Analysis, From (2026 to 2034)
Europe Adhesion Barrier Market Size
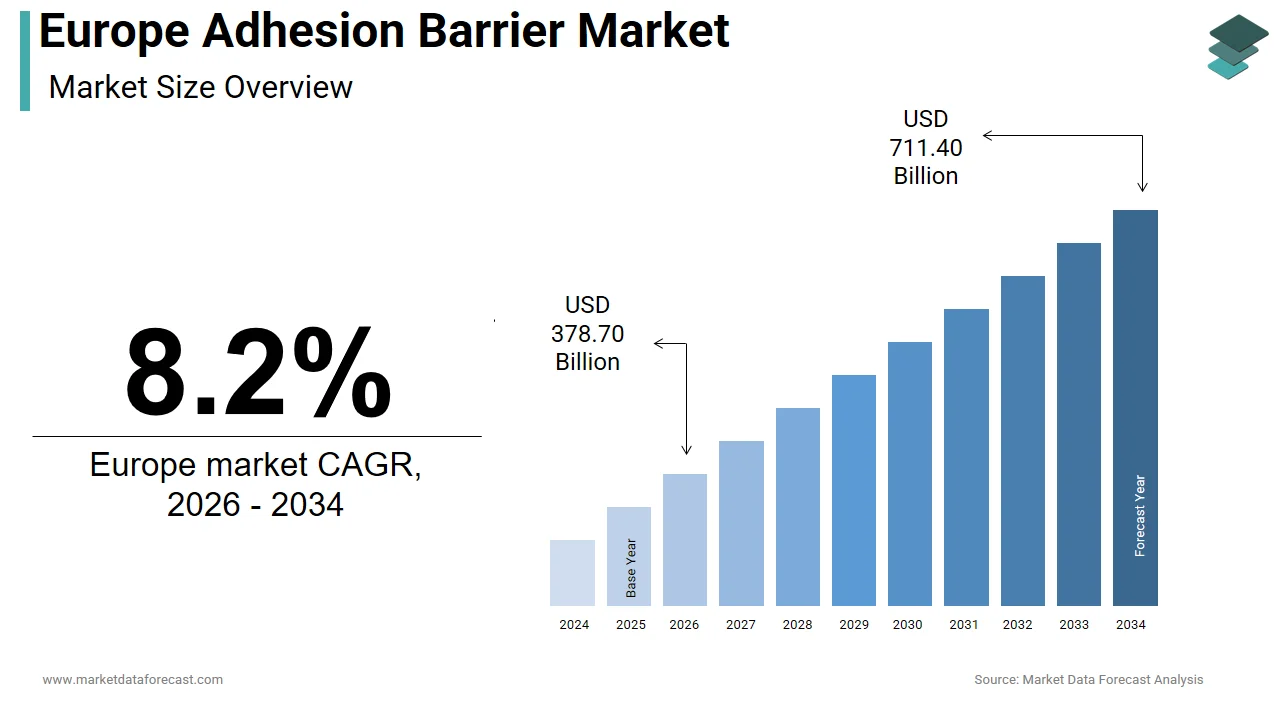
The size of the Europe adhesion barrier market was valued at USD 350 million in 2025. This market is expected to grow at a CAGR of 8.2% from 2026 to 2034 and be worth USD 711.40 million by 2034 from USD 378.70 million in 2026.
MARKET DRIVERS
The European adhesion barrier market is growing with the increasing number of surgical procedures. It is estimated that the growing awareness of the use of adhesion barriers after surgery in developing countries will drive the demand for adhesion barriers and hence contribute to the growth rate of the market during the forecast period.
The demand for adhesion barriers is growing due to the increasing prevalence of chronic diseases, the increase in the elderly population, and the increasing number of surgeries performed among the aging population. The emergence of companies focused on the R&D of adhesion barriers and the launch of new products and devices on the market are further accelerating the European market. In addition, it is believed that the growing awareness of the use of Adhesion Barriers after surgery in developing countries will drive the demand for Adhesion Barriers and hence contribute to the growth of the Adhesion Barriers market during the period of forecast 2023-2028.
MARKET RESTRAINTS
However, the risk of using adhesion barriers in patients with a history of hypersensitivity and the choice to use adhesion barriers among surgeons have been significant factors hampering the growth of the market. The market still faces barriers in terms of accessibility, lack of health education, and lack of awareness of the importance of treatment.
MARKET OPPORTUNITIES
Abdominal surgery is one of the most common causes of adhesions; it is estimated that about 75-85% of all adhesions are attributed to abdominal surgery. Most laparoscopic procedures cause adhesions. In Europe, 500,100 laparoscopic surgeries are performed each year. These factors are expected to have a positive impact on the growth of the market.
MARKET CHALLENGES
Regulatory requirements for medical device registration are complex and change with changing needs. These regulations are complicated, mainly because of the legal aspects associated with them. The quality and performance of adhesion barriers are necessary aspects that require rigorous evaluation to ensure public safety. Complex and strict standards are followed for the enrollment of medical devices. Strict regulatory requirements are challenging the growth of the Adhesion Barriers market for the forecast period (2020-2025). However, the limited applications of certain types of adhesion barrier products and the high cost of surgeries are expected to hamper the growth of the adhesion barriers market.
REPORT COVERAGE
| REPORT METRIC | DETAILS |
| Market Size Available | 2025 to 2034 |
| Base Year | 2025 |
| Forecast Period | 2026 to 2034 |
| Segments Covered | By Product, Product Form, Surgical Application, and Region. |
| Various Analyses Covered | Global, Regional and Country-Level Analysis, Segment-Level Analysis, Drivers, Restraints, Opportunities, Challenges; PESTLE Analysis; Porter’s Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
| Countries Covered | UK, France, Spain, Germany, Italy, Russia, Sweden, Denmark, Switzerland, Netherlands, Turkey, Czech Republic, and the Rest of Europe |
| Market Leaders Profiled | Baxter International Inc., Medtronic Plc., C. R. Bard, Inc., Johnson & Johnson, Sanofi Group, Atrium Medical Corporation, Integra Lifesciences Corporation, Anika Therapeutics Inc., FzioMed, Inc., and MAST Biosurgery AG.. |
REGIONAL ANALYSIS
Europe is expected to represent the second-largest share of the global adhesion barriers market due to the increasing use of advanced adhesion barriers and increasing product development by existing manufacturers.
The UK adhesion barriers market held the dominant share in the European region in terms of revenue in 2022 due to the developed medical infrastructure and favorable reimbursements for adhesion barriers.
Germany is expected to outperform all regions with the highest CAGR during the forecast period 2023-2028 due to the increase in the geriatric population and the increased incidence of cardiac surgery and neurosurgery that require surgical procedures. In addition, growing private sector investments in healthcare and rapidly expanding medical tourism are expected to boost the market in the region.
KEY MARKET PLAYERS
Key players operating in the Europe Adhesion Barrier Market profiled in this report are Baxter International Inc., Medtronic Plc., C. R. Bard, Inc., Johnson & Johnson, Sanofi Group, Atrium Medical Corporation, Integra Lifesciences Corporation, Anika Therapeutics Inc., FzioMed, Inc., and MAST Biosurgery AG.
MARKET SEGMENTATION
This Europe adhesion barrier market research report is segmented and sub-segmented into the following categories.
By Product
- Synthetic
- Natural
By Product Form
- Film
- Gel
- Liquid
By Surgical Application
- Gynaecological
- Cardiovascular
- Neurological
- Orthopaedic
- General/Abdominal
- Urological
- Reconstructive
By Country
- UK
- France
- Spain
- Germany
- Italy
- Russia
- Sweden
- Denmark
- Switzerland
- Netherlands
- Turkey
- Czech Republic
- Rest of Europe
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2000
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com