Latin America Biologics Market Size, Share, Trends & Growth Forecast Report By Source, Product, Biologics Manufacturing Type, Disease Category and Country (Brazil, Mexico, Argentina, Chile, Rest of Latin America) – Industry Analysis From 2025 to 2033.
Latin America Biologics Market Size
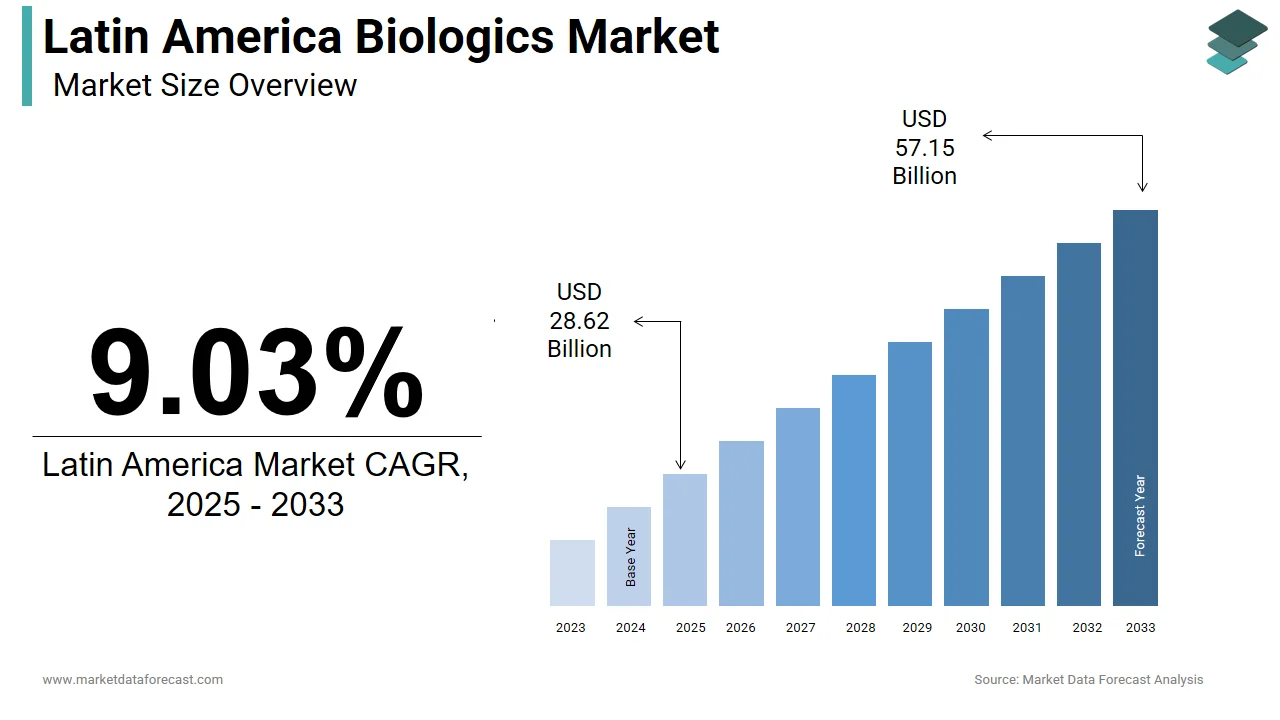
The size of the Latin America biologics market was valued at USD 26.25 billion in 2024. This market is expected to grow at a CAGR of 9.03% from 2025 to 2033 and be worth USD 57.15 billion by 2033 from USD 28.62 billion in 2025.
MARKET DRIVERS
Rising Chronic Diseases and Biologics Demand
The rising incidence of chronic diseases in Latin American countries, the growing number of COVID-19 cases, and the rising demand for biologics are majorly promoting the growth rate of the Latin American biologics market.
The Latin American biologics market is further expected to be driven by such factors as increased capital investment by major market players coupled with increased support and initiatives from governments of various countries around the world for the development and implementation of biologics, the increase in the prevalence of chronic diseases and the growth in demand and preference for innovative drugs.
Several new biotechnology companies receive funding from government agencies. Many companies are investing heavily in the development of organic products. Currently, more than half of the drug candidates at the discovery stage are biologics, such as proteins, peptides, and monoclonal antibodies. Biologics are expected to contribute about half of the revenue generated by the top 100 pharmaceuticals in the coming years. Emerging markets also offer room for innovation. Biopharmaceutical and biotechnology companies in these emerging markets are investing heavily in research to develop innovative molecules. The growing demand for corrective treatments for fatal diseases, such as cancer and diabetes, highlights the need for drug research and development. Hence, the demand for biosafety test kits for effective testing and quality control.
Additionally, factors such as the growing cancer burden, the rigorous efforts of key market players to develop novel biologic therapeutics, and growing investments for the R&D activities are expected to favor the market to grow further. Growth in the incidence of obesity and higher acceptability for the new biological therapies are factors that help the growth rate of the market climb further. The rising adoption of inactive lifestyle changes, increasing middle-class population, and rapid urbanization are expected to provide room for the market to grow.
MARKET RESTRAINTS
Stringent Regulations and Global Approval Complexities
Strict regulatory policies and heavy investments required for R&D are a few factors limiting the biologics market growth in this region. The biologics drugs are highly complicated and need to maintain and control throughout the development and production procedures; this concerned factor is expected to impede the market growth. Lack of awareness on biologics among physicians and specialists will inhibit the market growth to a small extent.
The management of the global life cycle of biologics, including biologic therapies and vaccines, is complex, and there is a lack of harmonization/alignment between drug regulatory agencies globally. Not only do regulations for post-approval changes vary globally, but the time it takes for the drug regulatory agency to review and approve changes also differs from country to country. This creates challenges for the regulated biopharmaceutical industry, as change cannot be implemented globally. This results in complexity for the manufacturer and the coexistence of "variants" of the product on the market. More importantly, this complexity translates into delayed access to innovation, increased drug costs, and, ultimately, supply constraints.
REPORT COVERAGE
| REPORT METRIC | DETAILS |
| Market Size Available | 2024 to 2033 |
| Base Year | 2024 |
| Forecast Period | 2025 to 2033 |
| Segments Covered | By Source, Product, Biologics Manufacturing, Disease Category, and Country. |
| Various Analyses Covered | Global, Regional and Country-Level Analysis, Segment-Level Analysis, Drivers, Restraints, Opportunities, Challenges; PESTLE Analysis; Porter’s Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
| Countries Covered | India, China, Japan, South Korea, Australia, New Zealand, Thailand, Malaysia, Vietnam, Philippines, Indonesia, Singapore, Rest of APAC |
| Market Leaders Profiled | Gilead Science, AbbVie, Merck & Co, Novo Nordisk, Biogen, Pfizer, CSL, Shire, Johnson & Johnson, Allergan Plc., Samsung biologics, Medly, and Astellas Pharma Inc. |
COUNTRY LEVEL ANALYSIS
Geographically, the biologics market in Latin America is fragmented, and only the largest countries, such as Argentina and Brazil, are attractive to international suppliers of organic products. Therefore, these leading countries are leading the manufacturing and marketing in the region.
Businesses have been drawn to the promise of expanding local economies in the first decade of this century, a skilled and cheap workforce, the rise of generics, strong coverage government regulation of drugs, and weak patent control (now in a more sophisticated and regularized era). But the strong local pharmaceutical industry has had to contend with large multinationals for many years, and in many cases, local companies have formed regional business groups. Local businesses, which historically only served markets in their home country, have consolidated and expanded to other Latin American countries.
Booming organic products industry in Latin America, with its own research, development, production, and marketing capabilities. The top three biologics-producing countries in Latin America are Argentina, Brazil, and Mexico. Due to its population and the size of the market, Brazil leads the biologics process.
KEY MARKET PLAYERS
Gilead Science, AbbVie, Merck & Co, Novo Nordisk, Biogen, Pfizer, CSL, Shire, Johnson & Johnson, Allergan Plc., Samsung biologics, Medly, and Astellas Pharma Inc. are some of the companies playing a crucial role in Latin America biologics market.
MARKET SEGMENTATION
This Latin America biologics market research report is segmented and sub-segmented into the following categories.
By Source
- Microbial
- Mammalian
- Others
By Product
- Monoclonal Antibodies
- Vaccines
- Recombinant Proteins
- Antisense, RNAi, & Molecular Therapy
- Others
By Biologics Manufacturing
- Outsourced
- In-house
By Disease Category
- Oncology
- Infectious Diseases
- Immunological Disorders
- Cardiovascular Disorders
- Hematological Disorders
- Others
By Country
- Brazil
- Mexico
- Argentina
- Chile
- Rest of Latin America
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 1600
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com