Middle East and Africa Adhesion Barrier Market Size, Share, Trends & Growth Forecast Report By Product, Product Form, Surgical Application and Country (KSA, UAE, Israel, rest of GCC countries, South Africa, Ethiopia, Kenya, Egypt, Sudan, rest of Middle East and Africa) – Industry Analysis, 2025 to 2033
Middle East and Africa Adhesion Barrier Market Size
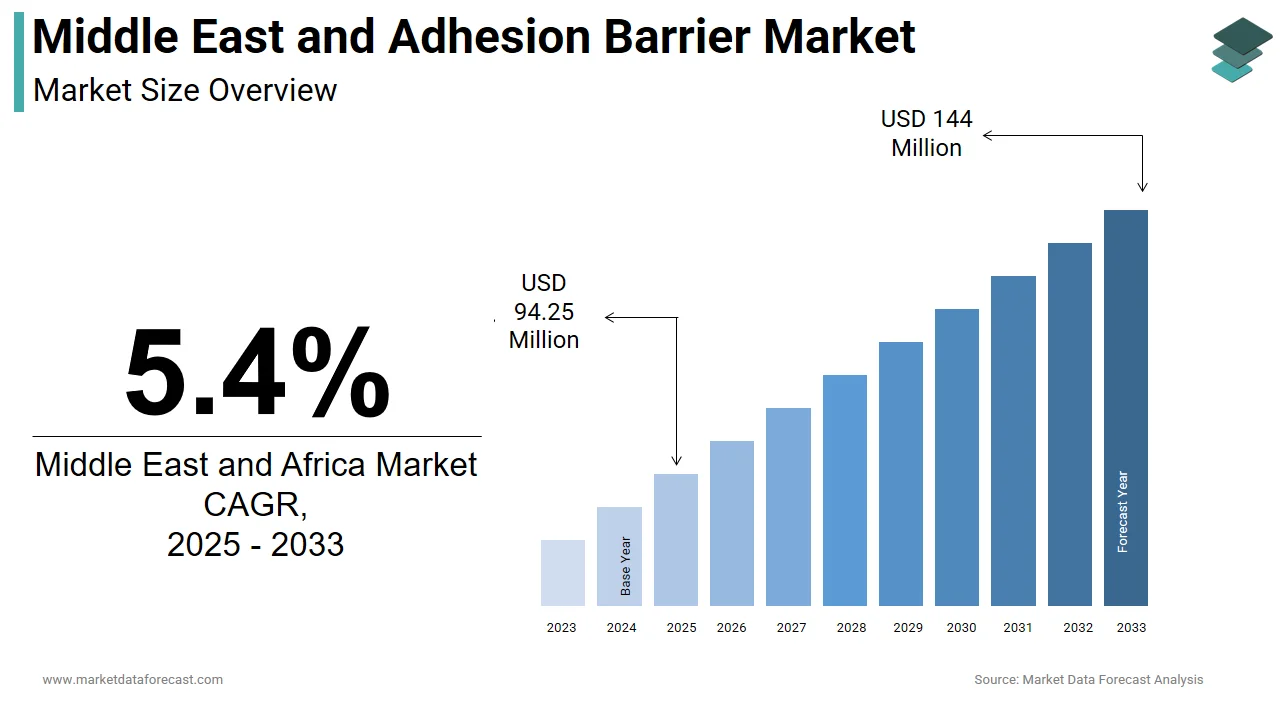
The Middle East and Africa adhesion barrier market was valued at USD 89.42 million in 2024, is estimated to reach USD 94.25 million in 2025, and is projected to reach USD 144 million by 2033, growing at a CAGR of 5.4% from 2025 to 2033.
MARKET DRIVERS
Y-O-Y growth in the incidence of Neurological, Gynecological, Ophthalmic, and Heart Disorders in the Middle East and Africa region is expected to drive the market growth. According to the World Heart Association, 7.6million people suffer from circulatory and heart disease. Heart and circulatory disease accounted for around 27% of all deaths in the Middle East regions in 2020. Therefore, treatment for these disorders requires surgery at times, increasing the demand for barriers to adherence.
In addition, the increase in healthcare expenditure associated with the increasing FDA approval for various adhesion barrier products is also increasing the market's growth. Moreover, favorable reimbursement policies and growing awareness of various surgical procedures drive market growth during the forecast period. Furthermore, the rapid growth of the geriatric population, the epidemiological shift from infectious diseases to chronic diseases, the increase in life expectancy, the expansion of private sector hospitals in rural areas, and the increased demand for surgical procedures drive the demand for surgical interventions.
Surgeons believe they can cost-effectively reduce the risk of postoperative complications in patients through adhesion barriers. Hence, these new advancements are expected to drive the Middle East and Africa adhesion barrier market growth in the forecasted period.
The growth of the Middle East and Africa adhesion barrier market is further expected to be driven by the increasing number of gynecological laparoscopic procedures. Moreover, the increasing adoption of gel form and film form in various gynecological procedures, including endometriosis surgery, ovarian cystectomy, and ectopic pregnancy, is driving the market's growth. The slow adoption of minimally invasive techniques in developing countries is expected to impact the adoption of barriers to membership positively. Minimally invasive surgeries require expensive equipment, such as cameras and robot-assisted devices. Adoption of these devices is expected to be moderate in developing countries due to price-sensitive customers.
MARKET RESTRAINTS
With the increased risk of postoperative adherence, the market still faces barriers in accessibility, lack of health education, and lack of awareness of the importance of treatment. Although surgeons are aware of these risks, they are reluctant to use adhesion barriers due to the lack/reliable and limited clinical evidence supporting the safety and efficacy of marketed adhesion barrier products. In addition, due to issues related to the safety and effectiveness of adhesion barriers, companies are having difficulty obtaining marketing authorizations for their products. Additionally, in recent years the market has seen the discontinuation of approved adhesion barrier products due to poor clinical outcomes.
The limited applications of certain types of adhesion barrier products and the high cost of major surgical procedures are some of the factors that are expected to hamper the market's growth.
REPORT COVERAGE
| REPORT METRIC | DETAILS |
| Market Size Available | 2024 to 2033 |
| Base Year | 2024 |
| Forecast Period | 2025 to 2033 |
| Segments Covered | By Product, Product Form, Surgical Application, and Region. |
| Various Analyses Covered | Global, Regional and Country-Level Analysis, Segment-Level Analysis, Drivers, Restraints, Opportunities, Challenges; PESTLE Analysis; Porter’s Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
| Countries Covered | KSA, UAE, Israel, rest of GCC countries, South Africa, Ethiopia, Kenya, Egypt, Sudan, rest of Middle East and Africa |
| Market Leaders Profiled | Baxter International Inc., Medtronic Plc., C. R. Bard, Inc., Johnson & Johnson, Sanofi Group, Atrium Medical Corporation, Integra Lifesciences Corporation, Anika Therapeutics Inc., FzioMed, Inc., and MAST Biosurgery AG. |
REGIONAL ANALYSIS
Geographically, the Middle East and Africa adhesion barrier market is expected to showcase a sluggish growth rate during the forecast period due to the increase in the geriatric population and the increase in the incidence of heart and neurosurgery, which require surgeries. In addition, growing private sector investments in healthcare and rapidly expanding medical tourism are expected to boost the market in the region. As a result, most of the regions of Saudi Arabia, Turkey, the United Arab Emirates, and Kuwait are expected to contribute to maximum growth of the region during the forecast period.
KEY MARKET PLAYERS
Baxter International Inc., Medtronic Plc., C. R. Bard, Inc., Johnson & Johnson, Sanofi Group, Atrium Medical Corporation, Integra Lifesciences Corporation, Anika Therapeutics Inc., FzioMed, Inc., and MAST Biosurgery AG are some of the companies playing an active role in the Middle East and Africa adhesion barrier market.
MARKET SEGMENTATION
This research report on the Middle East and Africa adhesion barrier market has been segmented and sub-segmented into the following categories:
By Product:
- Synthetic Adhesion Barriers
- Regenerated Cellulose
- Hyaluronic Acid
- Polythene Glycol
- Natural Adhesion Barriers
- Fibrin
- Collagen & Protein
By Product Form:
- Film Adhesion Barriers
- Gel Adhesion Barriers
- Liquid Adhesion Barriers
By Surgical Application:
- Gynecological Surgeries
- Cardiovascular Surgeries
- Neurological Surgeries
- Orthopedic Surgeries
- General/Abdominal Surgeries
- Urological Surgeries
- Reconstructive Surgeries
- Other Surgeries
By Country:
- KSA
- UAE
- Israel
- rest of GCC countries
- South Africa
- Ethiopia
- Kenya
- Egypt
- Sudan
- rest of Middle East and Africa
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 1600
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com