North America Adhesion Barrier Market Size, Share, Trends & Growth Forecast Report By Product, By Product Form, By Surgical Application and Country (United States, Canada, Mexico, Rest of North America) – Industry Analysis, 2024 to 2033
North America Adhesion Barrier Market Size
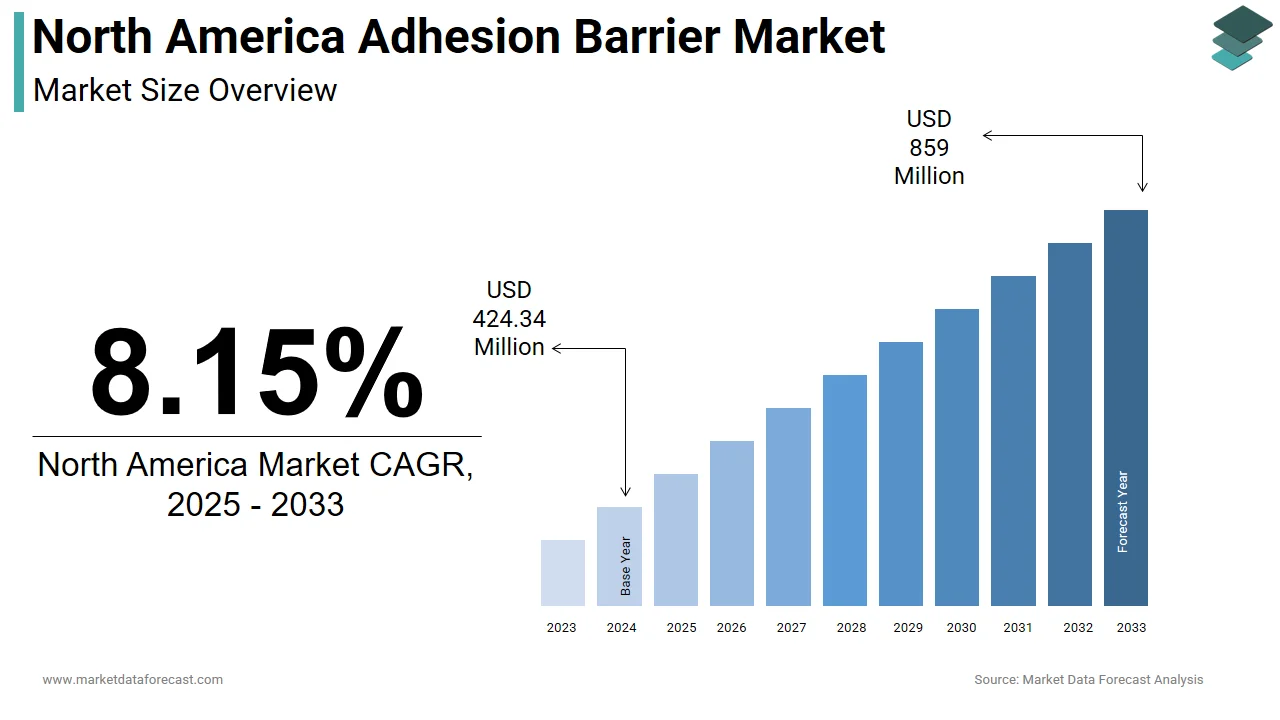
The North America adhesion barrier market was valued at USD 424.34 million in 2024 and is projected to reach USD 859 million by 2033, growing at a CAGR of 8.15% from 2024 to 2033.
MARKET DRIVERS
According to the National Cancer Institute, in 2019, approximately 1.75 million new cases of cancer were diagnosed in the United States. The increase in the geriatric population also significantly increases the use of adhesion barriers, as they suffer from various chronic diseases, increased incidence of gynecological disorders, neurological disorders, etc. heart disorders and ophthalmic disorders requiring surgery.
According to the World Health Organization (WHO), one in four people living in North America and Europe could be 65 or older by 2045. The number of people over age group 80 is expected to reach 428 million by 2050. In addition, with increasing urbanization and increasing inclination towards a healthy lifestyle, more people are expected to engage in various sporting activities. Hence, the result is an increase in sports-related injuries, which increases the use of anti-adhesion barriers. These factors play a key driving role in growth rate.
MARKET RESTRAINTS
On the other hand, due to the growing threat of adherence, after surgical treatment, the market still today sees barriers in terms of usability, a lack of awareness on the importance of the treatment as well as the need for health requirements, restraint market growth.
MARKET OPPORTUNITIES
The Covid-19 epidemic has posed a number of challenges for the health sector, weighing it down with a rapid number of cases. This has resulted in a reduction in the number of surgeries and, in turn, a weakening of the demand for adhesion barriers.
However, by mid-2022 the number of surgeries increased, leading to the sale of a variety of new opportunities for surgical tools and devices, such as adhesion barriers. This will cause better growth prospects for the adhesion barrier market in the region than in pre-pandemic business scenario.
MARKET CHALLENGES
Strict regulatory approvals and brand loyalty makes it difficult for new products to garner market sales, which can challenge the growth rate of the market.
REPORT COVERAGE
| REPORT METRIC | DETAILS |
| Market Size Available | 2024 to 2033 |
| Base Year | 2024 |
| Forecast Period | 2025 to 2033 |
| Segments Covered | By Product, Product Form, Surgical Application, and Region. |
| Various Analyses CoveredProduct, Test Type, Technique, End User | Global, Regional and Country-Level Analysis, Segment-Level Analysis, Drivers, Restraints, Opportunities, Challenges; PESTLE Analysis; Porter’s Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
| Countries Covered | United States, Canada, Mexico, Rest of North America |
| Market Leaders Profiled | Baxter International Inc., Medtronic Plc., C. R. Bard, Inc., Johnson & Johnson, Sanofi Group, Atrium Medical Corporation, Integra Lifesciences Corporation, Anika Therapeutics Inc., FzioMed, Inc., and MAST Biosurgery AG. |
REGIONAL ANALYSIS
The Americas are currently at the forefront of the global market and are expected to maintain their leadership position throughout the analysis period, given the strong presence of a massive group of medical device suppliers. The growing number of comprehensive research and development activities and the increase in laparoscopic procedures will also drive the demand for adhesion barriers in the years to come.
The increasing rate of surgeries in the region is well noted. Growth in the United States is driven by advancements in technology, older patients who expect higher levels of function, and reimbursement models that encourage complex fusion surgery.
Adhesion barriers are also useful in cancer surgeries. According to the American Cancer Society estimate, new cases of lung cancer in the United States for 2019 are expected to be around 2.35 million, and around 154,150 deaths are expected from lung cancer. The National Institutes of Health estimated that cancer treatment cost the United States a total of $ 150.5 billion in 2018, of which $ 14.4 billion was due to lung cancer. Surgeons believe they can cost-effectively reduce the risk of postoperative complications in patients through the use of adhesion barriers. Hence, these new advancements are expected to drive the market growth in North America.
KEY MARKET PLAYERS
Companies playing a dominant role in the North America Adhesion Barrier Market profiled in this report are Baxter International Inc., Medtronic Plc., C. R. Bard, Inc., Johnson & Johnson, Sanofi Group, Atrium Medical Corporation, Integra Lifesciences Corporation, Anika Therapeutics Inc., FzioMed, Inc., and MAST Biosurgery AG.
MARKET SEGMENTATION
This research report on the North America Adhesion Barrier Market has been segmented and sub-segmented into the following categories:
By Product
- Synthetic
- Natural
By Product Form
- Film
- Gel
- Liquid
By Surgical Application
- Gynecological
- Cardiovascular
- Neurological
- Orthopaedic
- General/Abdominal
- Urological
- Reconstructive
By Country
- U.S
- Canada
- Rest of North America
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2000
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com