North America Active Implantable Medical Devices Market Size, Share, Trends & Growth Forecast Report By Product and Country (United States, Canada, Mexico, Rest of North America) – Industry Analysis From 2025 to 2033.
North America Active Implantable Medical Devices Market Size
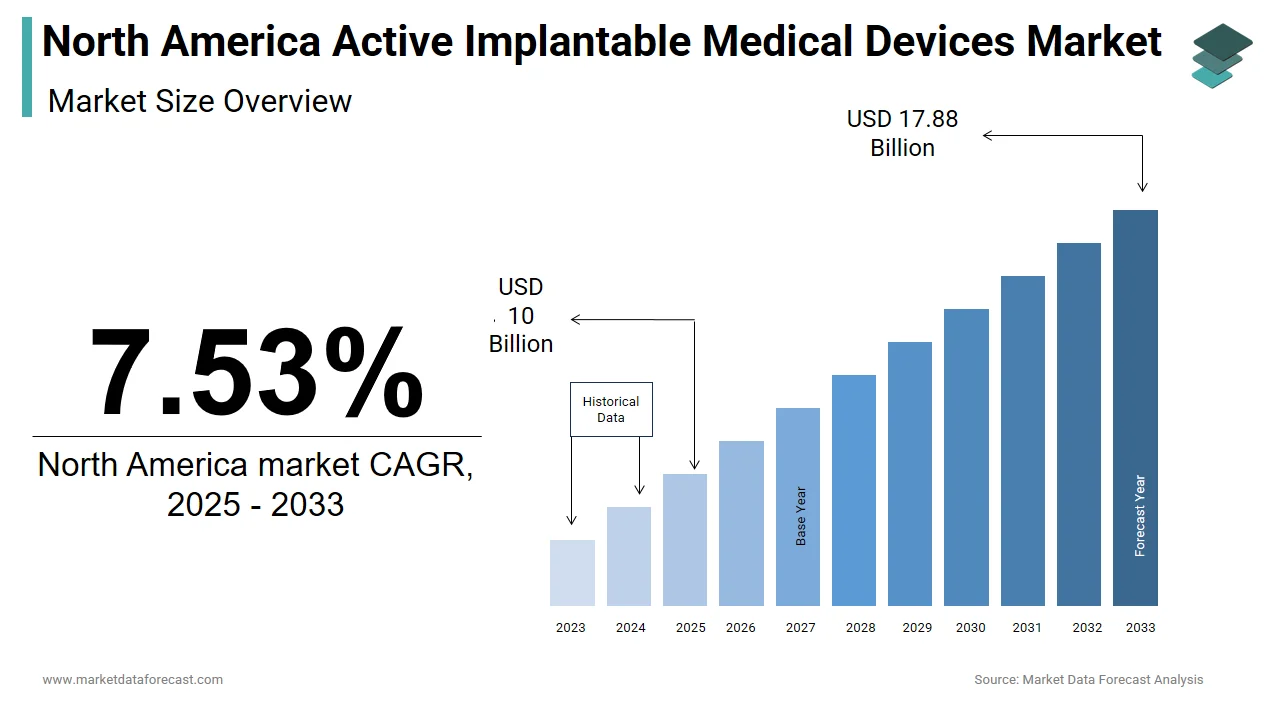
The size of the North America active implantable medical devices market was worth USD 9.30 billion in 2024. The North America market is anticipated to grow at a CAGR of 7.53% from 2025 to 2033 and be worth USD 17.88 billion by 2033 from USD 10 billion in 2025.
MARKET DRIVERS
The growth for active implantable medical devices in the North American region is growing due to the increasing prevalence of cardiovascular disease (CVD) and neurological disorders. CVD is primarily seen in the elderly, who are more susceptible to various ailments. The number of people aged 65 and over is expected to increase from 530 million in 2012 to nearly 1.65 billion in 2050, representing around 18% of the world's population. Implantable defibrillators and pacemakers are the primary medical devices used to treat various cardiovascular conditions, one of the leading causes of death across the globe. According to the World Health Organization, around 18 million people worldwide died from cardiovascular disease in 2014, and the numbers are expected to grow to 25 million by 2030. The increasing prevalence of cardiovascular diseases in Latin America has led to demand for technologically advanced products. According to the American College of Cardiology, CVD caused 900,000 deaths in the United States in 2019. Among Americans, an average person dies from cardiovascular disease every 45 seconds. These devices are placed near the heart and under the skin to act as artificial transvenous defibrillators and pacemakers etc., these are anticipated to help drive the market. The implantable pacemaker segment is growing rapidly, driven by increasing R&D funding for healthcare and increasing R&D trends in emerging economies of North America.
The increasing prevalence of chronic pain disorders and neuromuscular diseases, such as neuropathic pain, Parkinson's disease, Alzheimer's disease, diabetic neuropathy, etc., dramatically increases the demand for neurostimulator devices. The demand for such medical devices is anticipated to drive the market to new heights during the projected period. According to the Alzheimer's Association, approximately 5.5 million people aged 65 and over were thought to have Alzheimer's disease in 2016 in the United States. These figures are expected to reach 7.3 million in 2027 and around 13.9 million in 2050. Almost 15% of the population is over 65 and mainly needs these active implantable devices, such as deep brain stimulators. In addition, the high incidence of neurological disorders is fueling demand for neurostimulators, further boosting the active market for implantable medical devices in North America.
MARKET RESTARINTS
However, unfavorable reimbursement programs, issues with implantable medical devices, and the high cost of implants and associated surgeries may dampen the growth rate of the North American active implantable medical devices market.
REPORT COVERAGE
| REPORT METRIC | DETAILS |
| Market Size Available | 2024 to 2033 |
| Base Year | 2024 |
| Forecast Period | 2025 to 2033 |
| Segments Covered | By Product and Region. |
| Various Analyses Covered | Global, Regional and Country-Level Analysis, Segment-Level Analysis, Drivers, Restraints, Opportunities, Challenges; PESTLE Analysis; Porter’s Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
| Countries Covered | UK, France, Spain, Germany, Italy, Russia, Sweden, Denmark, Switzerland, Netherlands, Turkey, the Czech Republic, and the Rest of Europe. |
| Market Leaders Profiled | Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, BIOTRONIK SE & Co. KG, LivaNova PLC., Cochlear Limited, MED-EL, Sonova Holding AG, William Demant Holding A/S, and Nurotron Biotechnology Co. Ltd. |
GEOGRAPHICAL ANALYSIS
Based on region, North America held the largest share in the regional segment of the market. The large share of this regional segment can be credited to the growth in the geriatric population, the surge in the occurrence of cardiovascular and neurological diseases, high accessibility to advanced technologies, rise in the development of innovative and technologically advanced products, and the increase in the healthcare expenditure in the region.
In 2024, the United States dominated the North American active implantable medical devices market. The market is set to grow at a CAGR of 5%, attributed to the growth of the senior population, the increased prevalence of cardiovascular and neurological diseases, the high accessibility to advanced technologies, and the increased health spending in the region. According to the American Heart Association, approximately 5.8 million adults in the United States suffer from congestive heart failure. In addition, according to Parkinson's Disease Foundation, about 65,000 new cases of Parkinson's disease are reported annually in the United States. An increase in this disease will be responsible for the growth of the market.
However, Canada is expected to experience a healthy growth rate during the forecast period. The growth of this regional market can be attributed to the development of the senior population, the increasing incidence of chronic diseases, awareness about chronic diseases, favorable healthcare expenditure, the better healthcare infrastructure, growing competition with the United States in terms of market distribution, the availability of advanced technologies and less stringent regulations to develop high-end products and their approvals.
KEY MARKET PLAYERS
A few of the noteworthy companies operating in the North America Active Implantable Medical Devices Market profiled in the report are Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, BIOTRONIK SE & Co. KG, LivaNova PLC., Cochlear Limited, MED-EL, Sonova Holding AG, William Demant Holding A/S, and Nurotron Biotechnology Co. Ltd.
MARKET SEGMENTATION
This research report on the North America active implantable medical devices market is segmented and sub-segmented into the following categories.
By Product
- Implantable Cardioverter Defibrillators
- Transvenous Implantable Cardioverter Defibrillators
- Biventricular Implantable Cardioverter Defibrillators (ICDs)/Cardiac Resynchronization Therapy Defibrillators (CRT-Ds)
- Dual-Chamber Implantable Cardioverter Defibrillators
- Single-Chamber Implantable Cardioverter Defibrillators
- Subcutaneous Implantable Cardioverter Defibrillator
- Transvenous Implantable Cardioverter Defibrillators
- Implantable Cardiac Pacemakers
- Ventricular Assist Devices
- Implantable Heart Monitors/Insertable Loop Recorders
- Neurostimulators
- Spinal Cord Stimulators
- Deep Brain Stimulators
- Sacral Nerve Stimulators
- Vagus Nerve Stimulators
- Gastric Electrical Stimulators
- Implantable Hearing Devices
- Active Hearing Implants
- Passive Hearing Implants
By Country
- United States
- Canada
- Mexico
- Rest of North America
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2000
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com