Global Lyme Disease Diagnostics Market Size, Share, Trends & Growth Forecast Report By Diagnostic Tests, Diagnostic Technologies, End User and Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), Industry Analysis From 2025 To 2033.
Global Lyme Disease Diagnostics Market Size
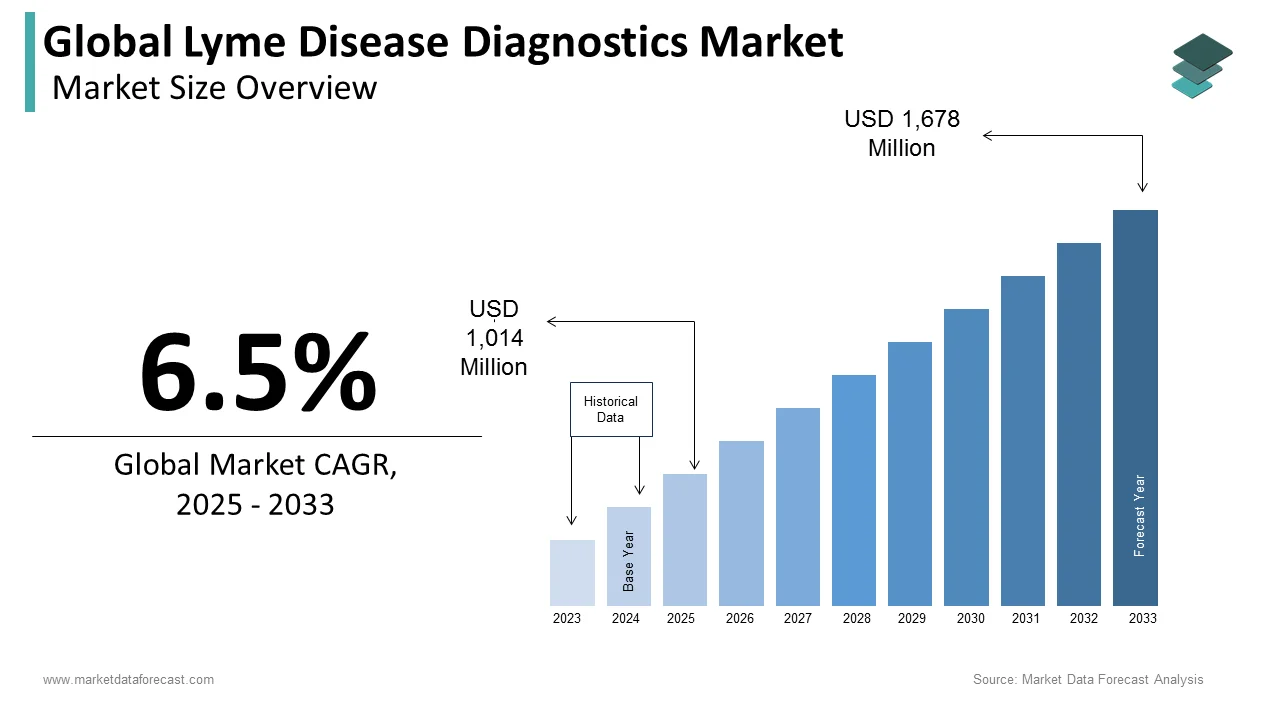
The size of the global lyme disease diagnostics market was worth USD 952 million in 2024. The global market is anticipated to grow at a CAGR of 6.5% from 2025 to 2033 and be worth USD 1,678 million by 2033 from USD 1,014 million in 2025.
Lyme disease is a bacterial infection caused by species borrelia; this disease generally spreads in humans through infected tick bites. Symptoms such as depression, fever, fatigue, and skin rashes result from infected people with the disease. It can be treated with antibiotics only if the condition is diagnosed at the early stages of its occurrence. However, if the disease is left untreated, it affects the central nervous system, heart, and joints. The diagnostic technology is used in different laboratories includes lymphocytic transformation tests, urine antigen tests, immunofluorescent staining, and others. The technologies are developing to diagnose Lyme diseases with the help of blood samples. According to the reports of the Centers for disease control and prevention, it is reported that the number of cases of tick-borne was 47,743 leading to the adoption of Lyme disease diagnostic tests.
MARKET DRIVERS
The global lyme disease diagnostics market is primarily driven by the increasing awareness about the novel techniques to diagnose lyme disease and identifying techniques of chronic Lyme infection from a blood sample.
The availability of these tests will improve the accuracy issue that plagued serology-based assays. The proliferation of Lyme disease is a key factor expected to drive the growth of the target market. Also, market participants' research and development activities are expected to boost the Lyme disease diagnostics market's growth potential.
Furthermore, the expanding government initiatives to upgrade medical healthcare facilities would aid the market's growth in the coming years. Besides, the rising number of tick-borne infectious illnesses, growing occurrences of novel forms of vector-borne diseases, and increasing healthcare spending are likely to propel the worldwide Lyme disease diagnostics market further. In partnership with the Centers for Disease Control and Prevention (CDC), the National Institute of Allergy and Infectious Diseases plays a critical role in supporting the development of innovative approaches to enhance Lyme disease screening. Moreover, NIAID grantees collaborate with the Centers for Disease Control and Prevention (CDC). However, leading market players are engaged in the research and development of novel tests for Lyme disease diagnosis, which is projected to help the global Lyme disease diagnostics market's growth. Many individuals with Lyme-like symptoms are misdiagnosed because current serological assays cannot reliably detect antibodies to all borrelia burgdorferi strains. As a result, there is a growing demand for improved illness diagnostic tests.
MARKET RESTRAINTS
However, global Lyme disease diagnostics market expansion is constrained by a lack of public awareness about the condition and ineffective Lyme disease testing in undeveloped and emerging countries. The lack of Lyme disease therapy in rural regions has slowed the Lyme disease diagnostics market over the period.
REPORT COVERAGE
|
REPORT METRIC |
DETAILS |
|
Market Size Available |
2024 to 2033 |
|
Base Year |
2024 |
|
Forecast Period |
2024 to 2033 |
|
Segments Covered |
By Diagnostic Tests, Diagnostic Technologies, End User and Region |
|
Various Analyses Covered |
Global, Regional & Country Level Analysis, Segment-Level Analysis, DROC, PESTLE Analysis, Porter’s Five Forces Analysis, Competitive Landscape, Analyst Overview on Investment Opportunities |
|
Regions Covered |
North America, Europe, APAC, Latin America, Middle East & Africa. |
|
Market Leaders Profiled |
Boulder Diagnostics, Immunetics Inc., GlaxoSmithKline Pharmaceuticals Ltd, Bio-Rad Laboratories, Alere Inc. (ALR), Roche Diagnostics International Ltd, Graphene Frontiers, Covance Inc., Affymetrix Inc., Siemens AG, Baxter International Inc., and Fresenius Medical Care AG & Co. |
SEGMENTAL ANALYSIS
By Diagnostic Tests Insights
Based on the diagnostic test, the serological test segment is predicted to dominate the global Lyme disease diagnostics market during the forecast period due to the frequent practice and routine tests for the laboratories and hospitals to diagnose Lyme disease.
The urine antigen tests segment is another lucrative segment in the global lyme disease diagnostics market and is estimated to register a healthy growth rate during the forecast period.
By Diagnostic Technologies Insights
Based on technologies, the molecular diagnostic segment was the market leader in the global lyme disease diagnostics market in 2020 and is estimated to continue its lead throughout the forecast period. Advanced laboratories for the disease tests and healthcare facilities' availability boost the molecular diagnostic segment during the forecast period.
By End User Insights
Based on the end-user, the hospitals and clinics segment held the majority share with the increasing CAGR over the forecast years, and it is expected to dominate the market. The available commercial test kits for detecting Lyme diseases are expected to propel the market growth over the forecast period.
REGIONAL ANALYSIS
Geographically, the North America Lyme disease diagnostics market is estimated to play the leading role in the global market during the forecast period and is majorly driven by the factors such as addressing the growing prevalence of Lyme diseases, identifying innovative ways of diagnosing Lyme disease, and improving healthcare facilities. Research and development activities on Lyme disease are anticipated to provide opportunities for the players in the North American market.
The European market is anticipated to be the second-largest regional market globally and is projected to grow at a promising rate during the forecast period. An increase in lyme disease prevalence, growing awareness among people regarding lyme diseases, and increasing investments are expected to drive the European market.
The Asia Pacific market is one of the fastest-growing regional markets globally and is estimated to grow at the highest CAGR during the forecast period. Factors such as the rising prevalence of lyme diseases, the emergence of new lyme diagnostic tests, and the increasing focus on developing healthcare facilities to provide better healthcare treatments for the people in this region are expected to propel the market growth. Rising disposable income in urban areas is also a factor driving the growth rate of the market.
The Middle East and Africa Lyme disease diagnostics market are expected to register a steady growth rate during the forecast period. The increasing prevalence of Lyme diseases and support by the government to improve healthcare facilities are anticipated to fuel the growth rate of the market in this region.
KEY MARKET PARTICIPANTS
Some of the prominent companies operating in the global Lyme disease diagnostics market profiled in the report are Boulder Diagnostics, Immunetics Inc., GlaxoSmithKline Pharmaceuticals Ltd, Bio-Rad Laboratories, Alere Inc. (ALR), Roche Diagnostics International Ltd, Graphene Frontiers, Covance Inc., Affymetrix Inc., Siemens AG, Baxter International Inc., and Fresenius Medical Care AG & Co.
MARKET SEGMENTATION
This market research report on the global lyme disease diagnostics market has been segmented and sub-segmented into the following categories.
By Diagnostic Tests
-
Darkfield Microscopy
-
Urine Antigen Tests
-
Lymphocytic Transformation Test
-
Serological Tests
-
Immunofluorescence Staining
-
PCR Tests
By Diagnostic Technologies
-
Molecular diagnostics
-
Monoclonal antibodies
-
Flow cytometry
-
Brain MRI
-
Spinal tap
-
Echocardiogram
-
Electrocardiogram
-
Microdroplets
By End User
-
hospitals/clinics
-
Pharmaceutical Industries
-
Pathology labs
-
Research Institutes
By Region
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
The Middle East and Africa
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2500
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com
