Global Pulmonary Drug Delivery Devices Market Size, Share, Trends & Growth Forecast Report By Product, Application, Distribution Channel and Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), Industry Analysis From 2025 To 2033.
Global Pulmonary Drug Delivery Devices Market Size
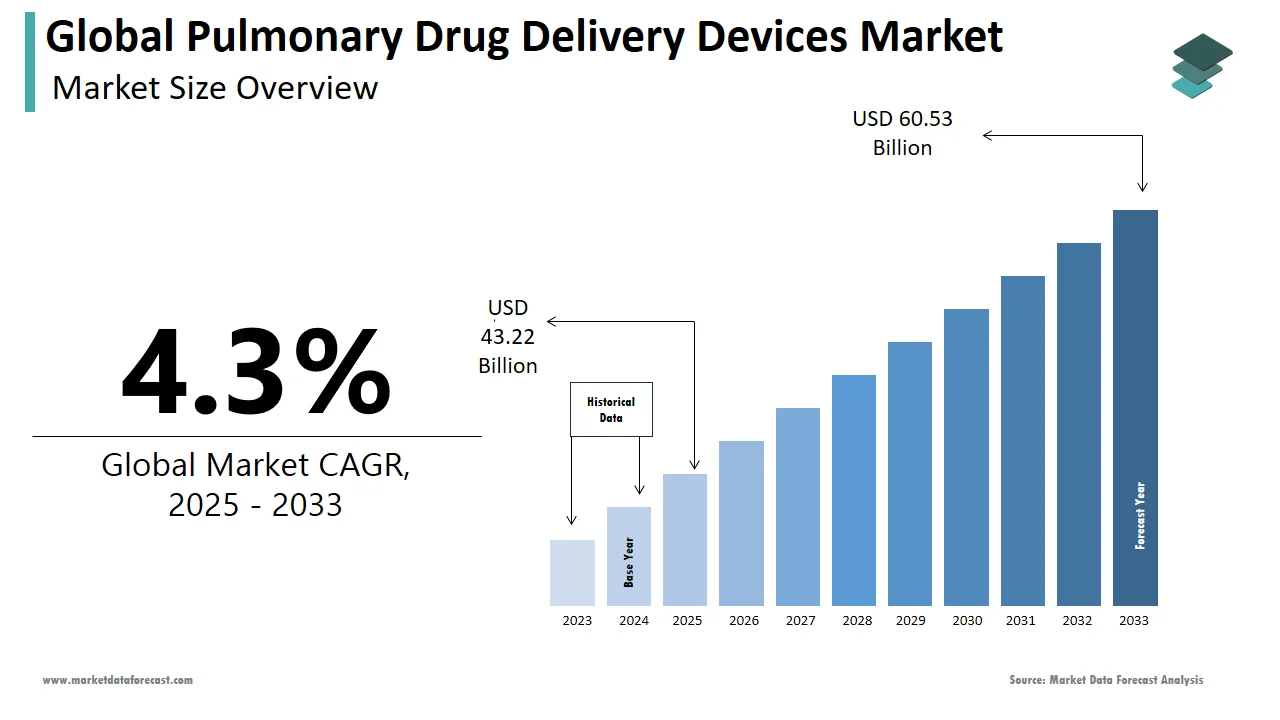
The size of the global pulmonary drug delivery devices market was worth USD 41.44 billion in 2024. The global market is anticipated to grow at a CAGR of 4.3% from 2025 to 2033 and be worth USD 60.53 billion by 2033 from USD 43.22 billion in 2025.
Pulmonary drug delivery devices are specific systems used to deliver drugs to the lungs to treat diseases of the respiratory tract. They are extensively monitored, as the process can be fatal if not performed correctly. This drug delivery method is often preferred because of its non-invasive nature and the fact that the process gives faster results due to speedier administration. The benefits of a large surface area for absorption, high blood circulation, and higher permeability rate have attracted many takers to the method.
MARKET DRIVERS
The growing geriatric population worldwide and the rising prevalence of chronic diseases such as tuberculosis, bronchitis, and chronic obstructive pulmonary disease have helped the pulmonary drug delivery devices market.
The global pulmonary drug delivery devices market is currently divided into nebulizers, dose inhalers, powder inhalers, etc. And these products today are readily available in hospitals, clinics, pharmacies, etc. However, the growing market for medical health and the ever-increasing possibilities in the online market for the same have helped the market reach out to a broader audience. Many attempts are also being made to make the policies more streamlined and more affordable to the government by investing in this industry's research. The limitations of other drug delivery systems compared to the benefits of the pulmonary drug delivery system, like the better penetrability of the drug, minimum dosage requirement, and growing technological advancements, have helped the global pulmonary drug delivery devices market grow worldwide scale.
MARKET RESTRAINTS
However, a few hindrances are a part of the global pulmonary drug delivery devices market. Its high prices, the need for medically trained supervisors, and many regulatory policies must be followed.
Impact Of COVID-19 On the Global Pulmonary Drug Delivery Devices Market
As of Feb 2021, Coronavirus has infected over 4.4 million people in 215 countries and territories, resulting in nearly 300,000 deaths. We estimate that the share of infectious diseases has increased due to the COVID-19 outbreak and that this segment will rise significantly during the forecast period. The drug delivery market has developed rapidly due to COVID-19, and it is projected to expand substantially between 2020 and 2021. In several nations, the COVID-19 outbreak has been growing exponentially. This has necessitated improved patient care and the availability of additional nutritional/immunity assistance devices, such as inhalers and nebulizers, to virus-affected patients. This has led to increased adoption of drugs and some of the devices for improving patient care. There are many drugs, devices, and vaccines in different clinical trials for treating COVID-19 that are expected to be released in the coming months/years under emergency use authorization guidelines. As a result, pulmonary drug delivery devices showed positive growth during the forecast period.
REPORT COVERAGE
|
REPORT METRIC |
DETAILS |
|
Market Size Available |
2024 to 2033 |
|
Base Year |
2024 |
|
Forecast Period |
2025 to 2033 |
|
Segments Analysed |
By Product, Application, Distribution Channel, and Region |
|
Various Analyses Covered |
Global, Regional & Country Level Analysis, Segment-Level Analysis, DROC, PESTLE Analysis, Porter’s Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
|
Regions Analysed |
North America, Europe, Asia Pacific, Latin America, the Middle East, and Africa |
|
Market Leaders Profiled |
AstraZeneca plc., Boehringer Ingelheim Gmbh, GlaxoSmithKline plc, Clement Clarke International, Briggs Healthcare, Novartis AG, Koninklijke Philips N.V., 3M Company, Actelion Pharmaceuticals Ltd, Aerogen, etc. |
SEGMENTAL ANALYSIS
By Product Insights
The Metered Dose Inhalers (MDI) segment had the highest market share of 66.7% of the worldwide market in 2022 based on the product. The high acceptance rate of these devices in developing regions such as the Asia Pacific and Latin America and the involvement of global key players in the production of novel MDIs contribute to the segment's revenue. The Dry Powder Inhaler (DPI) market is expected to rise at a healthy pace in the coming years. End users are likely to use these inhalers more due to benefits such as cost-effectiveness, ease of administration, and preparation time, among others.
By Application Insights
Based on the applications, chronic obstructive pulmonary disease is regarded as having the most substantial value in the market share in the forecast period. COPD's exponential rise can be attributed to the disease's high incidence and mortality rates. Revenue created by this segment is growing due to favorable reimbursement policies for COPD treatment and an increase in the number of government and non-government organizations that provide COPD treatment. The asthma segment had the highest market share. Asthma management is thought to begin with an effective inhalation therapy that involves DPIs and MDIs. For many people who have asthma, the demand for and use of asthma management devices such as MDIs and DPIs, boost this application's growth.
By Distribution Channel Insights
By distribution channel, the hospital pharmacies segment dominated the global pulmonary drug delivery device market. Patients prefer hospital pharmacies because they have extensive treatments, care, quick reimbursements, and insurance policies. Furthermore, the segment's growth is expected to be supported by a rise in hospitalizations. However, the e-commerce section is likely to expand with a remarkable growth rate in the coming years. Various factors, such as enhanced healthcare infrastructure in emerging regions and ease of purchase compared to retail or hospital pharmacies, have significantly helped the growth of this distribution channel.
REGIONAL ANALYSIS
The market for these pulmonary drug delivery devices has been established in developed regions like North America and Europe. North America accounted for the majority of PDDS in 2022, with a market value of USD 14.9 billion. The rising prevalence of diseases like COPD (Chronic Obstructive Pulmonary Disease) and asthma is likely to boost the growth rate of the market. As the number of patients affected by these diseases grows, so does the demand for diagnostic devices like PDDS (Pulmonary Drug Delivery Systems), boosting the region's growth. Furthermore, major pharmaceutical and medical device companies in the area and advanced healthcare infrastructure, an aging population, high disposable income, and lifestyle changes contribute to the market's overall growth in North America.
The Asia Pacific has soon been catching on owing to the growing focus on healthcare in this region. Due to the high prevalence of COPD, which indicates a significant socioeconomic burden in this area, the Asia Pacific region is expected to rise at an exponential rate of 6.2 percent. The continuous rise in the number of asthmatic and allergic rhinitis patients is another major factor expected to boost PDDS market demand in the Asia Pacific region over the forecast period. Additionally, an increase in the number of supporting non-governmental organizations, such as the Asia Pacific Association of Allergy, Asthma, and Clinical Immunology (APAAACI), is expected to raise patient awareness about respiratory treatment disorders, resulting in market development. The Asia Pacific region's growth is also expected to be aided by an increase in R&D activities by key market players to develop novel respiratory devices.
KEY MARKET PLAYERS
Companies leading the global pulmonary drug delivery devices market profiled in this report are AstraZeneca PLC, Boehringer Ingelheim Gmbh, GlaxoSmithKline plc, Clement Clarke International, Briggs Healthcare, Novartis AG, Koninklijke Philips N.V., 3M Company, and Actelion Pharmaceuticals Ltd.
RECENT MARKET HAPPENINGS
- In February 2020, IO Biotech announced the positive outcome of Phase 2 randomized trial interim analysis in first-line non-small cell lung cancer.
- In February 2020, Novartis announced a collaboration to develop a drug to treat Visceral Leishmaniasis.
- In February 2020, Novartis won EC approval for Anti-VEGF Treatment Beovu. It is a next-generation Anti-VEGF treatment for WET AMD, a leading cause of blindness throughout the world.
MARKET SEGMENTATION
This research report on the global pulmonary drug delivery devices market has been segmented and sub-segmented based on the product, application, distribution channel, and region.
By Product
- Dry Powder Inhalers
- Single-Dose Inhalers
- Multi-Dose Inhalers
- Metered Dose Inhalers
- Manually-Actuated Pressurized Inhaler
- Breath-Actuated Pressurized Inhaler
- Nebulizers
- Jet Nebulizers
- Ultrasonic Wave Nebulizers
- Vibrating Mesh Nebulizers
By Application
- Asthma
- Chronic Obstructive Pulmonary Disease (COPD)
- Cystic Fibrosis
- Others
By Distribution Channel
- Retail Pharmacies
- Hospital Pharmacies
- E-commerce
By Region
- North America
- The United States
- Canada
- Rest of North America
- Europe
- UK
- France
- Spain
- Germany
- Italy
- Rest of EU
- Asia Pacific
- India
- China
- Japan
- South Korea
- Australia & New Zealand
- Rest of APAC
- Latin America
- Brazil
- Mexico
- Argentina
- Chile
- Rest of LATAM
- Middle East
- Africa
Frequently Asked Questions
What is the current size of the pulmonary drug delivery devices market?
The global pulmonary drug delivery devices market is expected to be worth USD 41.44 billion in 2024.
What are the factors driving the growth of the pulmonary drug delivery devices market?
The growth of the pulmonary drug delivery devices market is driven by factors such as the increasing prevalence of respiratory diseases, the growing geriatric population, the rise in air pollution levels, and the increasing demand for home healthcare.
Who are the key players in the pulmonary drug delivery devices market?
Some of the key players in the pulmonary drug delivery devices market include GlaxoSmithKline plc, AstraZeneca plc, Boehringer Ingelheim GmbH, Merck & Co., Inc., Novartis International AG, Philips Respironics, Teva Pharmaceutical Industries Ltd., and others.
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2500
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com
