Global Influenza Market Size, Share, Trends & Growth Analysis Report – Segmented By Product Type (Vaccines & Drugs), Consumer (Children and Adults) and Region – Industry Forecast (2025 to 2033)
Global Influenza Market Size
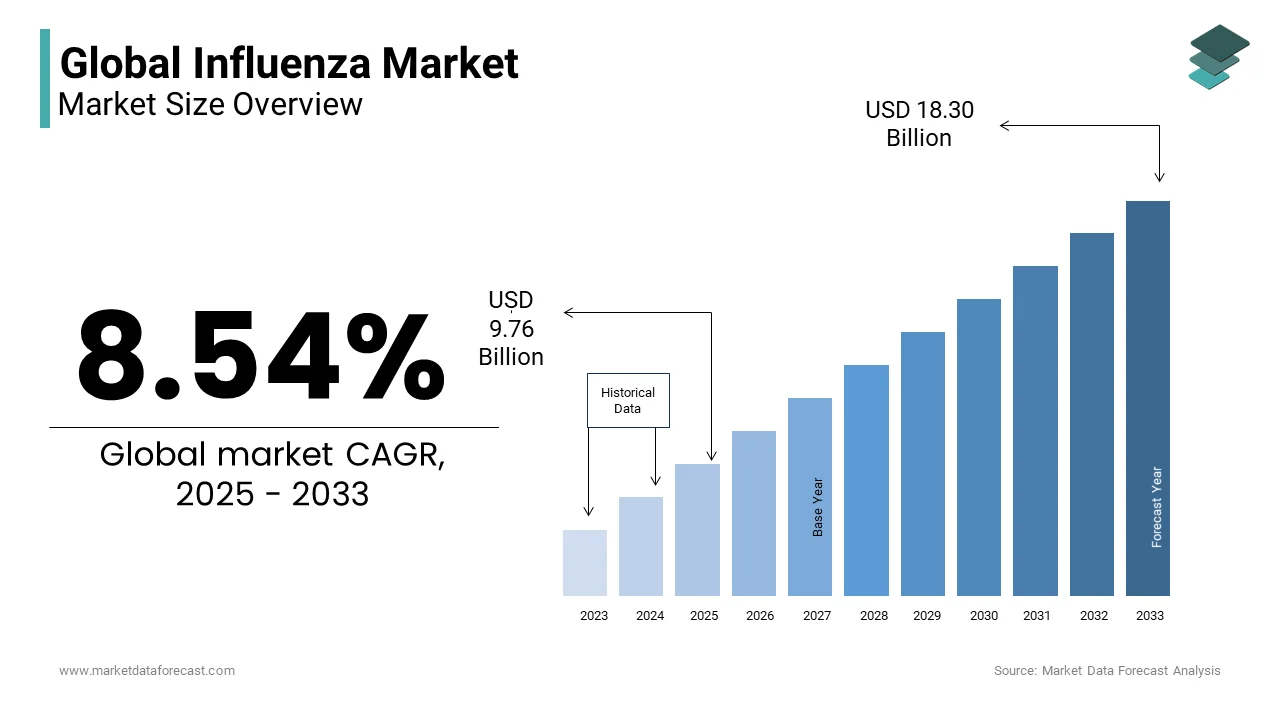
In 2024, the global Influenza Market was valued at USD 8.99 Billion and it is expected to reach USD 18.30 Billion by 2033 from USD 9.76 Billion in 2025, growing at a CAGR of 8.54% during the forecast period.
Influenza is a flu or viral infection which affects the respiratory system. The person affected by influenza may suffer from symptoms such as headaches, fever, fatigue, cough, muscle aches, congestion, and a running nose. There are four types of influenza viruses, which are type A, B, C, and D. These viruses travel through the respiratory system and transmit through direct contact or by taking virus-infected air particles. Therefore, influenza can be ideally prevented by vaccination.
MARKET DRIVERS
The growing focus of governments to provide better medical solutions for the people, increasing vaccine research, and low risk of side effects associated with influenza vaccines are a few factors accelerating the global influenza market growth.
According to the Centers for Disease Prevention and Control (CDC) estimates, an estimated 390,000 hospitalizations were recorded between 2019 and 2020. Increased funding for R&D activities, the emergence of novel vaccines, and the increased medical tourism prevalence fuel the influenza market's growth rate. Furthermore, the increased number of immunization programs at regular intervals and the availability of various influenza vaccines that the world health organization has prequalified are anticipated to favor the market. In addition, many influenza vaccines for treating the influenza A virus in animals are developing, and clinical trials are supposed to be launched over time.
Influenza vaccines are being modified specially for seasons to tackle the virus strains, which alter each season, motivating the market participants to focus more on innovative vaccines to handle all the infections and viruses and is expected to boost the market growth.
MARKET RESTRAINTS
The key factors that challenge the growth of the global influenza market are the time required to produce the vaccine and the manufacturing costs associated with very high development.
REPORT COVERAGE
|
REPORT METRIC |
DETAILS |
|
Market Size Available |
2024 to 2033 |
|
Base Year |
2024 |
|
Forecast Period |
2025 to 2033 |
|
CAGR |
8.54% |
|
Segments Covered |
By Product Type, Consumer, and Region |
|
Various Analyses Covered |
Global, Regional & Country Level Analysis, Segment-Level Analysis; DROC, PESTLE Analysis, Porter's Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
|
Regions Covered |
North America, Europe, APAC, Latin America, Middle East & Africa |
|
Market Leaders Profiled |
Sanofi-Pasteur (U.S.), AstraZeneca (United Kingdom), F. Hoffmann-La Roche (Switzerland), Novartis (Switzerland), GlaxoSmithKline (U.K.), Pfizer Inc. (U.S.), Merck & Co. (U.S.), Abbott Laboratories (U.S.), Mitsubishi Tanabe Pharma Corporation (Japan) and CSL Limited (Australia) |
SEGMENTAL ANALSIS
By Product Type Insights
Based on product, the vaccines segment is estimated to register a major share of the global influenza market during the forecast period owing to the rising demand for seasonal influenza vaccination to guarantee excellent protection against the deadly pandemic. In addition, global healthcare organizations such as the Centers for Disease Control and Prevention (CDC) have suggested baloxavir marboxil (Xofluza) vaccines to treat the symptoms in patients due to their significant health consequences.
By Consumer Insights
Based on the consumers, the children segment is the most lucrative and is projected to account for the most significant share of the global influenza market during the forecast period. This dominance is due to greater vaccine doses given to neonates, babies, and children to protect them against influenza illness from an early age. The high prevalence of influenza infection and its frequency among young children has prompted a variety of methods. In addition, according to the CDC, flu-related hospitalizations among children have boosted the need for childhood vaccinations in the newborn age group.
On the other hand, adult vaccination is now required to reduce the number of hospitalizations and fatalities caused by influenza. Due to the government's increased vaccination efforts and high vaccine dosage procurement by GAVI, PAHO, and UNICEF, the adult vaccines segment is expected to have a substantial share in this market during the forecast period.
REGIONAL ANALYSIS
Geographically, the North American influenza market held a significant share of the global market in 2022 and is expected to continue the trend throughout the forecast period owing to the factors such as the increased focus of governments on immunization programs, improvements in vaccine administration, increased incidence of influenza in the region, and the influx of new vaccines. North America is expected to dominate the global influenza market during the forecast period because certain companies are concentrating more on manufacturing innovative influenza vaccines for therapeutic purposes in countries like the United States. Factors such as rising influenza prevalence and the number of influenza screening tests conducted in this area propel the North American market forward throughout the forecast period. The rapid development of efficient influenza vaccines and technologically advanced vaccine production processes are expected to drive regional market growth. In addition, growing influenza incidence and high vaccine sales are expected to drive market growth in North America.
The European influenza market accounted for the second-largest share of the global market in 2022 and is also projected to see substantial growth during the forecast period owing to the expansion of vaccine coverage for high-risk populations, resulting in greater immunization rates among the European population. Additionally, growing product launches in this region are expected to boost adoption rates.
The Asia-Pacific influenza market is anticipated to grow at a healthy growth rate during the forecast period. Emerging economies and the increased adoption rate of influenza are supporting market growth.
Due to the improvements in healthcare facilities, increased disposable money, and rapid economic growth, the Latin American influenza market is anticipated to become a profitable market for influenza vaccinations. Latin America is another major contributor to the global market and is expected to grow rapidly during the forecast period due to increased disposable income, advancements in healthcare facilities, and escalated growth of economies.
The influenza market in the Middle East and Africa is anticipated to grow at a moderate CAGR during the forecast period.
KEYMARKET PLAYERS
Some of the promising companies leading the global influenza market profiled in this report are Sanofi-Pasteur (U.S.), AstraZeneca (United Kingdom), F. Hoffmann-La Roche (Switzerland), Novartis (Switzerland), GlaxoSmithKline (U.K.), Pfizer Inc. (U.S.), Merck & Co. (U.S.), Abbott Laboratories (U.S.), Mitsubishi Tanabe Pharma Corporation (Japan) and CSL Limited (Australia).
RECENT HAPPENINGS IN THIS MARKET
-
ResVax vaccine, a vaccine developed by Novavax Company for RSV diseases, has completed phase 3 of the trial process, and the outcomes were announced at the IDSOG meeting in August 2019.
-
Emergent BioSolutions Company developed a vaccine called NuThrax anthrax vaccine, for which the company has enrolled patients to begin the phase 3 trial of clinical study in 2018. The vaccine is a combination of anthrax and BioThrax vaccine developed by Emergent, which improves the body's immune response.
MARKET SEGMENTATION
This research report on the global influenza market has been segmented and sub-segmented based on product type, consumer, and region.
By Product Type
- Vaccines
- Drugs
By Consumer
- Children
- Adults
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Frequently Asked Questions
Which region accounted for the leading share in the global influenza market in 2025?
North America accounted for the most dominant share of the global market in 2025.
Which are a few of the notable players in the global influenza market?
Sanofi-Pasteur (U.S.), AstraZeneca (United Kingdom), F. Hoffmann-La Roche (Switzerland), Novartis (Switzerland), GlaxoSmithKline (U.K.), Pfizer Inc. (U.S.), Merck & Co. (U.S.), Abbott Laboratories (U.S.), Mitsubishi Tanabe Pharma Corporation (Japan) and CSL Limited (Australia) are some of the prominent companies in the influenza market.
Which segment by product dominated the influenza market in 2025?
Based on the product, the vaccine segment had the major share in the global influenza market in 2025.
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2500
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com
