Global Zika Virus Vaccines Market Size, Share, Trends & Growth Forecast Report By Type (Therapeutic Vaccines, Preventive Vaccines), End-user and Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), Industry Analysis From 2026 to 2034.
Global Zika Virus Vaccines Market Size
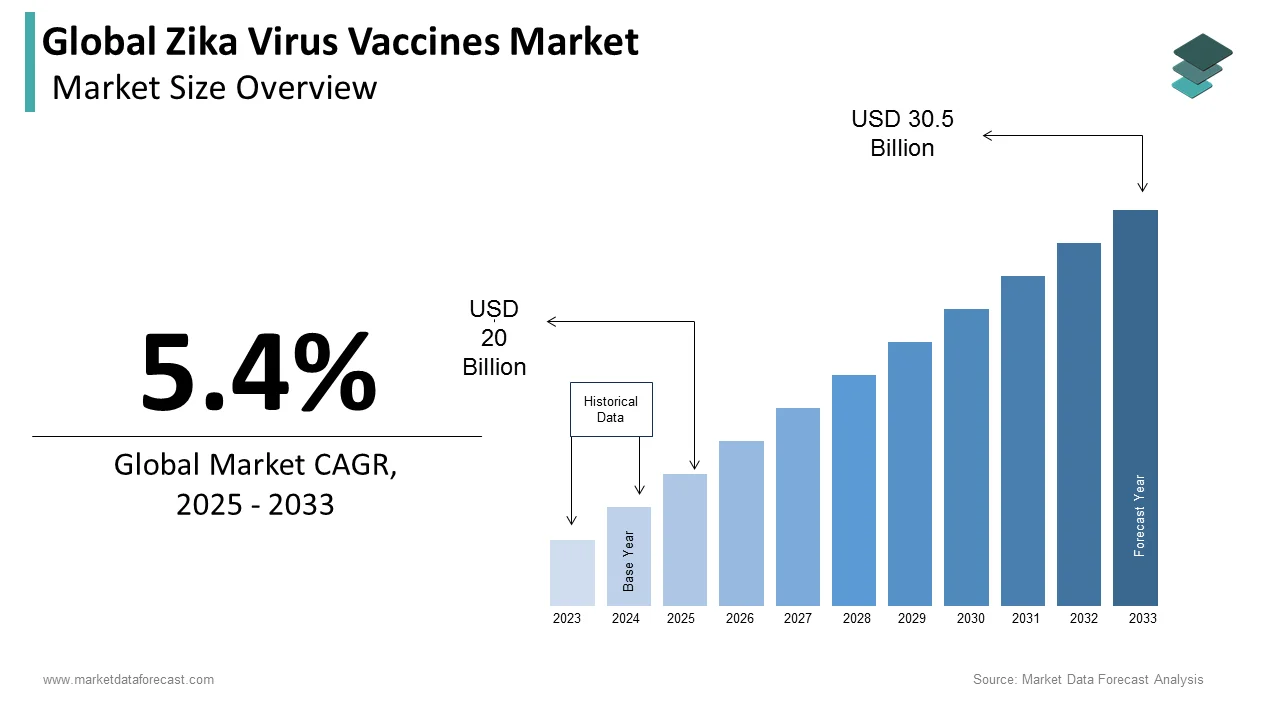
The global zika virus vaccines market was worth US$ 20 billion in 2025 and is anticipated to reach a valuation of US$ 32.11 billion by 2034 from US$ 21.08 billion in 2026, and it is predicted to register a CAGR of 5.4% during the forecast period 2026 to 2034.
MARKET DRIVERS
The growing incidence of the Zika virus globally, especially in regions with favorable mosquito vectors, is one of the major factors propelling the market growth.
Zika virus is a mosquito-borne viral infection. The impact of the Zika virus has reached various parts of the world due to its association with severe birth defects and neurological disorders. As per the data published by the World Health Organization (WHO), 91 countries and territories have experienced the transmission of the Zika virus since 2007. The increasing incidence of the Zika virus has fuelled awareness among people and healthcare professionals regarding the risks of the infection, especially for pregnant women and their unborn children. This has resulted in the increased demand for effective preventive measures such as the development and adoption of Zika virus vaccines. The urgency to combat the spread of the Zika virus has fuelled the demand for Zika virus vaccines and this trend is likely to continue in the coming years and drive the market growth.
The growing public health awareness and an increasing number of initiatives from governments further contribute to the Zika virus vaccines market growth.
The awareness of the risks and consequences of Zika virus infections among people has increased considerably in recent years. On the other hand, governmental organizations and public health organizations have been taking several measures to control and limit the transmission of the Zika virus. The governmental organizations of some countries have been offering financial support and research grants to support the R&D of Zika virus vaccines. Such factors drive the development and adoption of Zika virus vaccines and support market growth.
The supportive reimbursements from governments, technological advancements, rising focus on immunization, increasing healthcare expenditure, growing disposable income and a large population of virus-affected people propel the Zika virus vaccines market growth. Various vaccines are being developed to protect against viruses, such as mRNA, DNA-based, genetically engineered and inactivated vaccines. With the rise in zika virus incidences, many pharmaceutical and biotechnology companies are investing in this market. In a very rare case, the zika virus is also transmitted through blood transfusion. In 2016, the World Health Organization (WHO) declared that the major causes of the zika virus were asymptomatic.
The growing number of collaborations and partnerships between governments, pharmaceutical companies, research institutions, and non-profit organizations to support the development and production of Zika virus vaccines, the rising trend of international travel and tourism and increasing awareness of the risks of Zika virus to pregnant women favor the market growth. The growing support from regulatory bodies such as implementing fast-track pathways, streamlined regulatory processes and expedited reviews, increasing number of awareness campaigns and initiatives by international organizations such as the World Health Organization (WHO) to combat the spread of Zika virus and the potential for future Zika virus outbreaks further boost the market’s growth rate.
MARKET RESTRAINTS
The scientific and technical challenges associated with the development of safe and effective Zika virus vaccines majorly hamper the market growth.
Stringent assessments of the safety, efficacy and quality of Zika virus vaccines by the regulatory bodies challenge the market growth. High costs associated with the development of Zika virus vaccines, vaccine hesitancy or reluctance among the public and the presence of other preventive measures such as vector control, public health education, and personal protection measures further impede the market growth.
REPORT COVERAGE
| REPORT METRIC | DETAILS |
| Market Size Available | 2025 to 2034 |
| Base Year | 2025 |
| Forecast Period | 2026 to 2034 |
| Segments Covered | By Application, Type, Dermatoscopes, and Region. |
| Various Analyses Covered | Global, Regional and Country-Level Analysis, Segment-Level Analysis, Drivers, Restraints, Opportunities, Challenges; PESTLE Analysis; Porter’s Five Forces Analysis, Competitive Landscape, Analyst Overview of Investment Opportunities |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Market Leaders Profiled | Takeda Pharmaceutical Co. Ltd., NewLink Genetics Co., Immunovaccine Inc., GeneOne Life Science Inc., GlaxoSmithKline PLC, Inovio Pharmaceuticals, Inc., Bharat Biotech International Ltd., Hawaii Biotech Inc., and Sanofi S.A., and Others. |
SEGMENTAL ANALYSIS
By Type Insights
The therapeutic vaccines segment is anticipated to grow with the fastest CAGR in the global market for zika virus vaccines during the forecast period. The growing number of Zika virus infections worldwide creates a strong demand for therapeutic vaccines and is driving the growth of therapeutic vaccines. The potential of therapeutic vaccines in reducing the severity of symptoms and complications associated with Zika virus infection and the growing need to address the current lack of specific antiviral treatments for Zika virus infection further boost the segment’s growth rate.
The preventive vaccines segment is anticipated to account for a promising share of the global market during the forecast period. The efforts from governments and public health organizations worldwide to promote vaccination as a preventive measure against Zika virus infection are one of the key factors propelling the segmental growth. The growing need for preventive vaccines to protect pregnant women and their unborn children from Zika virus infection and the rising demand for preventive vaccines among travelers and individuals residing in or traveling to Zika-affected areas further boost the growth rate of the segment.
By End-User Insights
The hospital segment held the largest share of the global market in 2025 and this trend is projected to continue throughout the forecast period. The growing healthcare infrastructure, especially in developing regions, supporting the administration and distribution of Zika virus vaccines in hospitals, the availability of vaccines in hospitals, and collaborations of hospitals with pharmaceutical companies and research institutions to conduct clinical trials and vaccine distribution programs drive the growth of the hospital segment.
The clinic segment had the second-largest share of the global market in 2025 and is predicted to grow at a steady CAGR during the forecast period. The convenience and proximity of clinics, the emphasis of clinics on preventive care and vaccination programs and targeted outreach programs, such as community health campaigns and educational initiatives by clinics contribute to the growth of the clinics segment.
REGIONAL ANALYSIS
Latin America Zika Virus Vaccines Market Analysis
Latin America dominated the worldwide Zika virus vaccine market in 2025 and this trend is projected to continue over the forecast period. The region's supremacy can be ascribed to the rising number of persons infected with the Zika virus in countries like Brazil, Bolivia, and Peru and the presence of key players. The rise in Zika-infected people is expected to spur effective vaccinations to reduce infection rates. This increases the level of preparedness in the event of future Zika outbreaks. As a result, it can boost Zika vaccine demand and provide opportunities for critical companies over the forecast period. Zika fever has infected the populations of 69 countries, causing mild disease, and has spread over Central and South America.
Asia Pacific Zika Virus Vaccines Market Analysis
The Asia Pacific regional market is predicted to grow rapidly during the forecast period. The high population density and favorable mosquito vectors in the Asia-Pacific region majorly drive the growth of the APAC market. Rising healthcare expenditure in countries across the Asia-Pacific region, growing focus on public health initiatives and immunization programs and the implementation of proactive Middle East and Africasures to combat Zika virus transmission by the governments of APAC countries further drive the APAC market growth.
Middle East and Africa Zika Virus Vaccines Market Analysis
The Middle East and Africa market is anticipated to hold a considerable share of the global market during the forecast period owing to the growing investments in healthcare infrastructure, improving access to immunization programs, emerging outbreaks and international collaborations with research organizations and pharmaceutical companies to address public health challenges to promote the Middle East and Africa market growth.
KEY MARKET PLAYERS
Some of the most prominent companies leading the global zika virus vaccines market profiled in this report are
- Takeda Pharmaceutical Co. Ltd.
- NewLink Genetics Co.
- Immunovaccine Inc.
- GeneOne Life Science Inc.
- GlaxoSmithKline PLC
- Inovio Pharmaceuticals, Inc.
- Bharat Biotech International Ltd.
- Hawaii Biotech Inc.
- Sanofi S.A.
GLOBAL ZIKA VIRUS VACCINES MARKET NEWS
- In June 2016, the FDA approved the first human clinical trial for GLS–5700, an Inovio Pharmaceuticals zika vaccine being produced. The distribution of phase I-IV clinical trials suggests that most Zika virus trials were in phase I. In 2017, the NIAID Vaccine Research Center (VRC) accepted a phase 2 clinical studies grant for a DNA vaccine.
- Sanofi S.A initiated a vaccine research and development initiative in early 2016 with the release of Dengvaxia (Dengue vaccine) to help the company identify and develop an effective vaccine against the Zika virus.
- In early 2016, the WHO introduced a global public awareness program through its Strategic Response Program and Joint Strategy for Zika. This strategy outlines all the issues at the time of infections to be taken care of. Further, in mid-2016, by publishing Zika Risk Assessment for the European Region, the WHO Regional Office for Europe established a learning program to raise awareness.
MARKET SEGMENTATION
This research report on the global zika virus vaccines market has been segmented and sub-segmented based on type, end-user, and region.
By Type
- Therapeutic Vaccines
- Preventive Vaccines
By End-User
- Hospitals
- Clinics
- Research Institutes
- Others
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- The Middle East and Africa
Frequently Asked Questions
1. What is the global zika virus vaccines market?
The global zika virus vaccines market involves development and distribution of vaccines to prevent zika virus infection and its related complications globally
2. What drives growth in the global zika virus vaccines market?
Growth is driven by rising zika outbreaks, government funding, advances in mRNA and DNA vaccines, and increasing awareness of congenital zika syndrome risks
3. Which vaccine types are most common in the global zika virus vaccines market?
mRNA, DNA, and inactivated vaccines are leading formulations in development and deployment in the global zika virus vaccines market
4. How do vaccine platforms impact the global zika virus vaccines market?
Innovations in vaccine platforms improve efficacy, reduce side effects, and accelerate development, boosting the global zika virus vaccines market
5. What regions have highest demand in the global zika virus vaccines market?
Tropical regions in South America, Southeast Asia, and Africa drive demand due to endemic presence and outbreak risks in the global zika virus vaccines market
6. Who are key players in the global zika virus vaccines market?
Companies like Sanofi, Pfizer, Bharat Biotech, Moderna, and Inovio lead vaccine development and commercialization globally
7. How do government initiatives influence the global zika virus vaccines market?
Government grants and public health campaigns support R&D and immunization, accelerating market growth worldwide
8. What challenges affect the global zika virus vaccines market?
Challenges include virus mutation, regulatory hurdles, vaccine hesitancy, and logistical distribution issues in the global zika virus vaccines market
9. How does vaccine efficacy affect the global zika virus vaccines market?
High efficacy builds public trust and regulatory approval, essential for widespread adoption in the global zika virus vaccines market
10. How important is vaccine safety in the global zika virus vaccines market?
Safety profiles determine regulatory approvals and public acceptance critical to success in the global zika virus vaccines market
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2500
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com